Clinical effectiveness analisis of LMWH in the prevention of deep vein thrombosis and pulmonary embolism
-
Copyright
© 2013 PRO MEDICINA Foundation, Published by PRO MEDICINA Foundation
User License
The journal provides published content under the terms of the Creative Commons 4.0 Attribution-International Non-Commercial Use (CC BY-NC 4.0) license.
Authors
Background: The aim of the study was to assess the clinical effectiveness of low molecular weight heparins in prevention of deep vein thrombosis and pulmonary embolism in comparison with physical methods, unfractionated heparin and placebo, by a systematic review of reports in medical literature.
Methods: The assessment of the clinical effectiveness of undertaken interventions was compliant with the principles of systematic review (EBM), based on the Cochrane Collaboration guidelines. Statistical analysis and meta-analysis were performed by means of the RevMan 4.2 software version.
Results: Regarding the risk of postoperative overall deep-vein thrombosis and proximal deep-vein thrombosis, a meta-analysis of obtained results revealed a trend towards low-molecular-weight heparin versus the results of physical methods. However, the difference between the analyzed groups did not reach statistical significance.
Compared to placebo, the results of deep vein thrombosis risk assessment by meta-analysis showed statistically significant differences in favor of low-molecular-weight heparins (RR = 0.50, 95% CI: 0.34, 0.74, P = 0.0004, NNT = 23).
In comparison to the group, receiving unfractionated heparin, the observed differences did not attain statistical significance, neither in thromboembolism prevention nor in deep vein thrombosis treatment.
Regarding the risk of any bleeding episodes, the meta-analysis showed a statistically significant difference in favor of low-molecular-weight heparins administered in the study group vs. placebo results in the control group (RR = 1.55, 95% CI: 1.07, 2.24, P = 0.02) with the NNH equal to 94.
Conclusion:Low molecular weight heparins are effective and safe treatment for venous thromboembolism versus placebo, however, no statistically significant advantages were observed vs. physical methods or unfractionated heparins.
Introduction
Low molecular weight heparins are used with increasing frequency in the primary prevention and treatment of venous thrombosis and acute myocardial infarction. Low molecular weight heparins (LMWH) are more expensive than unfractionated heparin but associated with additional benefits, such as shorter hospitalization and the possibility of treatment at home.
Due to the increasing popularity of LMWG and the relatively high public reimbursement, allocated for this group of drugs, there are more and more questions about the cost-effectiveness of such procedures. This analysis provides some basis for consideration of the advisability of using low molecular weight heparins. Based on meta-analyses of available clinical evidence, an assessment was conducted of the clinical effects of low molecular weight heparins versus placebo, unfractionated heparin or physical methods.
Clinical picture and epidemiology of venous thromboembolism
The definition of venous thromboembolism includes two diseases: Deep Vein Thrombosis (DVT) and Pulmonary Embolism (PE), which is often a complication of the former.
Deep vein thrombosis and related complications – pulmonary embolism and the post-thrombotic syndrome – form a very serious interdisciplinary problem of today’s medicine, with various risks which may result from these complications. Pulmonary embolism is a severe, life-threatening disease, and the post-thrombotic syndrome, a chronic condition – is often the cause of permanent disability [47].
According to the Polish data, deep vein thrombosis affects about 50 thousand people per year and pulmonary embolism of varying severity is identified in about 20 thousand people,. being the cause of about 10% of all hospital deaths and a leader among preventable causes of mortality. Frequently, deep vein thrombosis has an occult clinical course. It can occur in hospitalized patients, as well as in apparently healthy individuals at any time during their life. Pulmonary embolism is often the first and final sign of deep vein thrombosis. The majority of unrecognized cases of thrombosis lead to the thrombotic syndrome and incidents of pulmonary embolism, later followed by chronic pulmonary hypertension [48].
The treatment of venous thromboembolism complications is extremely expensive – the costs are comparable to expenditures in oncology, arising not only from the treatment of acute thrombosis or early complications, but also from treatment of the post-thrombotic syndrome and pulmonary hypertension. Indirect costs are associated with days on sick-leave and paid sickness benefits.
Prevention of venous thromboembolism
Physical methods (intermittent pneumatic compression): The aim of physical methods is to reduce venous stasis in the legs, a major contributor to thrombosis formation. These methods are easy, and require relatively cheap measures, while being proven as fairly effective for patients with a moderate risk of thrombotic events. However, in cases of high risk of thrombosis, the outcomes are not satisfactory.
Safety is a great advantage of the physical methods, especially where the risk of bleeding complications, associated with the use of anticoagulants, is unacceptable, for example, after neurosurgical procedures, multiple accidental trauma or surgery within the eyeball.
Pharmacological methods: Pharmacological methods rely on the drugs that inhibit blood clotting. Despite a long list of available products, the medicinal products, most readily used in the prophylaxis of venous thromboembolism, are heparin and oral anticoagulants. Unfractionated heparin (UFH), administered subcutaneously and in small doses (5000 IU every 8-12 hrs.) is a standard method to prevent venous thromboembolism in patients with moderate and high risk of thrombosis. Low molecular weight heparins, administered by subcutaneous injection in small doses, demonstrate a significantly higher bioavailability (> 90%) vs. unfractionated heparin (20-30%). They also present a longer half-life and may be used in single daily doses. They do not require laboratory monitoring of their anticoagulant activity, due to their improved pharmacokinetic properties.
Other pharmacological therapies include oral anticoagulants, dextran, heparinoids and specific inhibitors of enzymes.
Methods
The search strategy was based on the Evidence Based Medicine principles, with the following electronic databases:
- The Cochrane Controlled Trials Register (CENTRAL)
- Cochrane Database of Systematic Reviews (CDSR)
- Medline (PubMed)
- Embase
- BioMed Central
- and medical electronic portals:
- BIOSIS Previews
- CINAHL Database
- PsycINFO
- European Public Assessment Report (EPAR)
- Health Canada
- Netherlands Pharmacovigilance Centre Lareb
- The Uppsala Monitoring Centre, and
- Thompson Micromedex
Additionally, to find more reliable data, a secondary search was carried out (systematic reviews and meta-analyses) in medical databases and existing independent HTA reports, available on the websites of institutions, cooperating with the Agency for Health Technology Assessment: International Network of Agencies for Health Technology Assessment (INAHTA), Health Technology Assessment International (HTAi) and the Centre for Reviews and Dissemination (CRD).
Scientific papers were also sought in sources other than medical information databases: in bibliographies of published literature reviews and references, used in clinical research publications, reports and abstracts from scientific conferences and clinical trial registries. Clinical experts were also invited to consult. Additional information was obtained by manual search of selected journals, the use of search engines and by contacts with authors of clinical trials.
Date from the last search of medical databases: 10 September 2007
The decision issue was defined according to the PICOS pattern (population, intervention, comparator, outcomes, study design) (Table 1).
Table 1. Issues vs key words in literature review
|
Issue |
Key words |
|
Clinical problem, population |
(#1) venous thromboembolism (#2) deep vein thrombosis (#3) pulmonary embolism |
|
Intervention |
(#4) low molecular weight heparin (#5) enoxaparin (#6) nadroparin (fraxiparin) (#7) dalteparin |
|
Comparators |
(#8) mechanical devices (#9) placebo (#10) unfractioned heparin |
|
Outcomes |
(#11) thromboprophylaxis (#12) VTE, DVT prophylaxis (#13) VTE, DVT prevention (#14) adverse event (#14) bleeding complication, risk of haemorrhage |
|
Study design |
(#15) randomized controlled trial (#16) randomized clinical trial (#16) RCT (#17) clinical trial |
Results
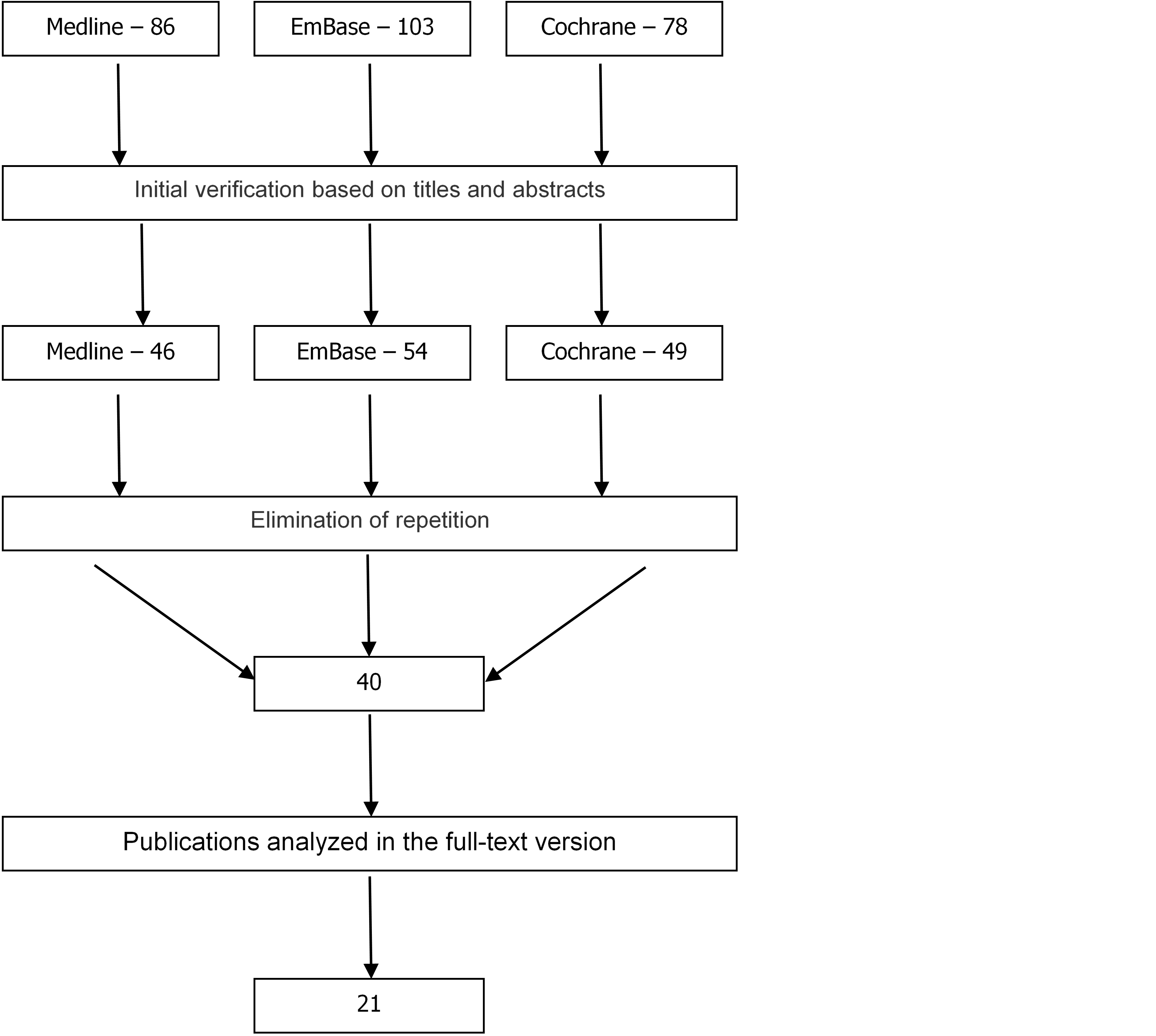
In result of searching medical databases, 267 publications were found on the use of low molecular weight heparins in prevention of venous thromboembolism (Figure1).
Initially, 40 publications were selected with data meeting inclusion criteria. Full texts of scientific reports were analysed to assess their reliability, providing, 21 publications, out of the original set of randomized clinical trials, which met the criteria and were eligible for later analysis in compliance with predefined assumptions.
Additionally, four secondary studies were found, being meta-analyses of the clinical efficacy and safety of low molecular weight heparins in prevention and treatment of venous thromboembolism, as compared with the physical methods, placebo or unfractionated heparin.
At all stages, the selection was made independently by two analysts. In any case of disagreement in verification, based on full text analysis of scientific reports, a final position was attained by consensus.
Meta-analysis results
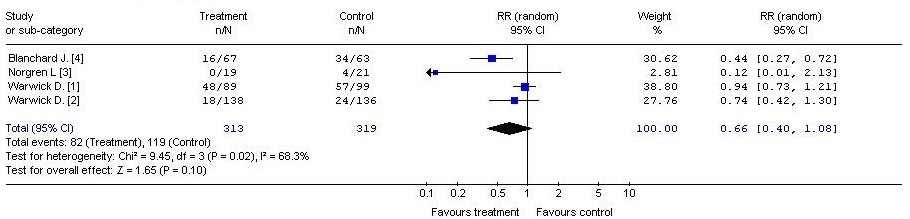
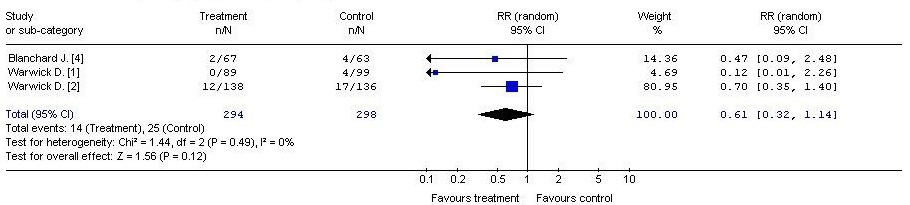
LMVH vs physical methods (Fig. 2, Fig. 3)
Treatment – LMWH
Control – physical methods (foot pump)
Outcome – overall episodes of deep vein thrombosis
Treatment – LMWH
Control – physical methods (foot pump)
Outcome – episodes of proximal deep vein thrombosis
With regard to the risk of postoperative deep vein thrombosis in both the total number of cases and the number of cases with proximal deep vein thrombosis, meta-analysis studies [1,2,3,4] showed a trend in favour of low molecular weight heparins versus the physical methods (intermittent pneumatic compression). The difference between the analyzed groups, however, did not reach statistical significance (RR = 0.66, 95% CI: 0.40, 1.08, p = 0.10). (RR = 0.61, 95% CI: 0.32, 1.14, p = 0.12) (Table 2).
Table 2. Episodes of deep vein thrombosis vs. episodes of proximal deep vein thrombosis in metaanalysis studies
|
Measured endpoint |
Number of studies |
Patients % (LMWH) |
Patient % (foot pump) |
RR [95% CI] |
GRADE score |
|
Episodes of deep vein thrombosis |
4 |
26.2% |
37.3% |
0.66 [0.40; 1.08] NS |
High |
|
Episodes of proximal deep vein thrombosis |
3 |
4.8% |
8.4% |
0.61 [0.32, 1.14] NS |
High |
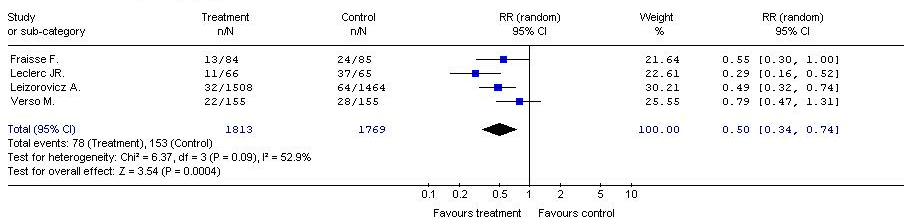
LMVH vs placebo (Fig. 4, Fig.5, Fig.6, Fig.7)
Treatment – LMWH
Control - placebo
Outcome – episodes of deep vein thrombosis
With regard to the risk of deep vein thrombosis, meta-analysis results of four primary clinical trials showed a statistically significant difference between the benefits of low molecular weight heparins in the study group vs. the placebo-treated control group. (RR = 0.50, 95% CI: 0.34, 0.74, p = 0.0004). The NNT (the Number Needed to Treat) was 23, which means that the administration of low molecular weight heparins instead of placebo to 23 patients, during the period of follow-up, was associated with avoiding deep vein thrombosis in one of them.
Treatment – LMWH
Control – placebo
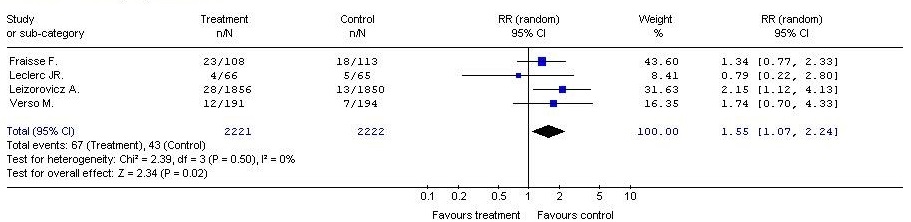
Outcome – any bleeding episodes
Treatment – LMWH
Control – placebo
Outcome – major bleeding episodes
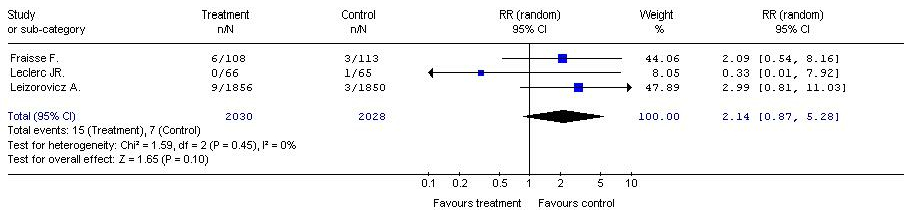
Treatment – LMWH
Control – placebo
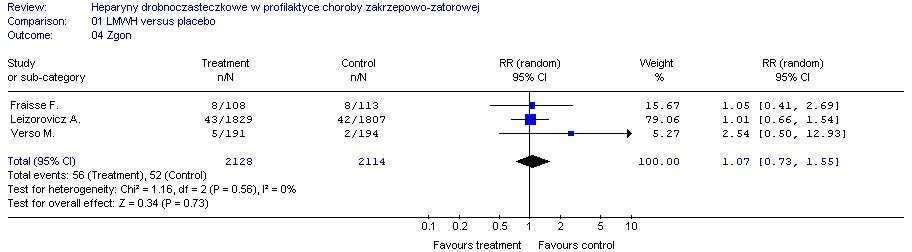
Outcome – death
Regarding the risk of any incidents of bleeding, the meta-analysis showed a statistically significant difference in favour of low molecular weight heparins, administered in the study group and compared to placebo in the control group (RR = 1.55, 95% CI: 1.07, 2.24, P = 0.02).
The NNH parameter (the Number Needed to Harm) was 94, which means that the administration of LMWH instead of placebo to 94 patients during the follow-up period was associated with bleeding events in one of them.
With regard to the risk of death from any cause, and clinically significant bleeding, the meta-analysis showed no statistically significant differences between the compared groups (RR = 2.14, 95% CI: 0.87, 5.28, P = 0.10); (RR = 1.55, 95% CI: 073, 1.55, P = 0.73) (Table 3).
Table 3. Risk of any incidents of bleeding (LMWH vs. placebo)
|
Measured endpoint |
Number of studies |
Patient % (LMWH) |
Patient % (placebo) |
RR [95% CI] NNT/NNH* |
GRADE score |
|
Episodes of deep vein thrombosis |
4 |
4,3% |
8,6% |
0,50 [0,34, 0,74] NNT = 23 |
High |
|
Minor bleeding episodes |
4 |
3% |
1,9% |
1,55 [1,07, 2,24] NNH = 94 |
High |
|
Major bleeding episodes |
4 |
0,7% |
0,3% |
2,14 [0,87, 5,28] NS |
High |
|
Death |
4 |
2,6% |
2,5% |
1,07 [0,73, 1,55] NS |
High |
* NNT and NNH parameters were calculated for statistically significant differences between the compared groups
** not significant
The meta-analysis showed a 50% decrease in the risk of deep venous thrombosis after low molecular weight heparins, compared to placebo, and NNT = 23. Only one study of pulmonary embolism did not confirm the statistically significant activity of LMWH versus placebo. There were no significant differences between the groups regarding the risk of clinically significant bleeding, which indicates an acceptable safety profile of low molecular weight heparins. Their high antithrombotic efficacy is much higher than any of the risks of bleeding events.
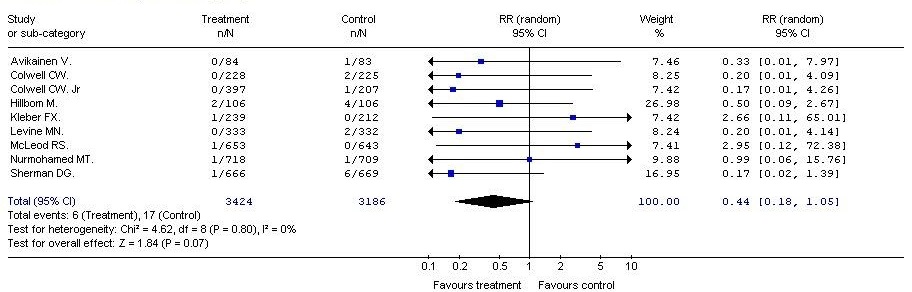
LMVH vs. UFH (Fig. 8, Fig. 9, Fig.10, Fig.11)
Treatment – LMWH
Control - UFH
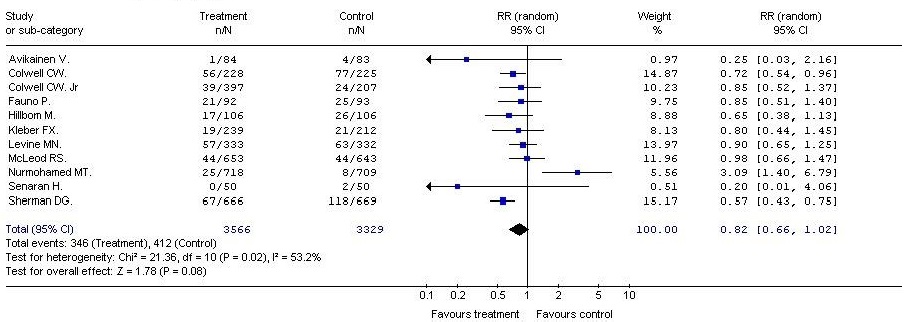
Outcome – episodes of deep vein thrombosis
Treatment – LMWH
Control - UFH
Outcome – episodes of pulmonary embolism
A meta-analysis of clinical efficacy showed a trend in favor in favor of LMWHs vs UFH in the prevention of thromboembolism. However, differences in the incidence of deep vein thrombosis and pulmonary embolism, did not achieve statistical significance (RR = 0.82, 95% CI: 0.66, 1.02, p = 0.08); (RR = 0.44, 95% CI: 0.18, 1.05, p = 0.07).
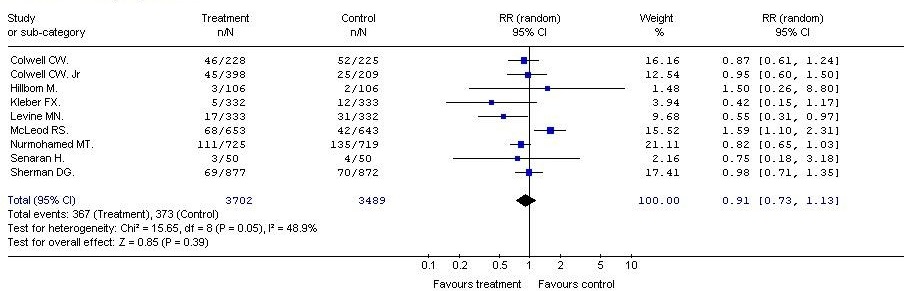
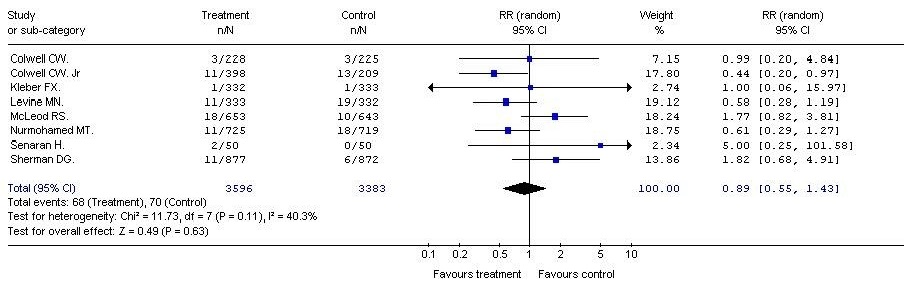
Treatment – LMWH
Control – UFH
Outcome – any bleeding episodes
Treatment – LMWH
Control – UFH
Outcome – major bleeding episodes – clinically significant
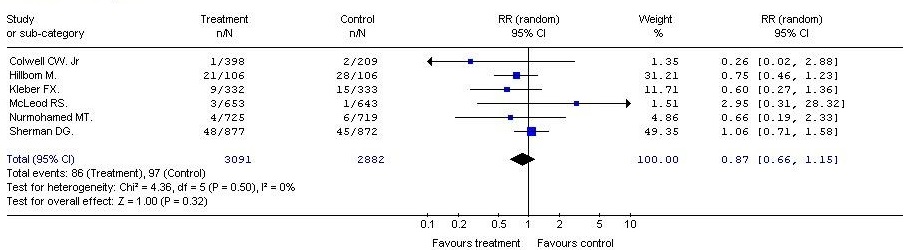
Treatment – LMWH
Control – UFH
Outcome – death
In all the measured safety parameters after LMWH vs. UFH, the risk of minor and major bleeding and the mortality rates were slightly lower in the groups, receiving low molecular weight heparins, compared to the control group. However, the differences between the analyzed therapeutic options were not statistically significant. (RR=0.91; 95% CI: 0.73, 1.13; p=0.39); (RR = 0.89; 95% CI: 0.55, 1.43; p = 0.63); (RR = 0.87; 95% CI: 0.66, 1.15; p = 0.32) (Table 4).
Table 4. Risk of minor and major bleeding and the mortality rates (LMWH vs. UFH)
|
Measured endpoint |
Number of studies |
Patient % (LMWH) |
Patient % |
RR [95% CI] NNT/NNH* |
GRADE score |
|
Episodes of deep vein thrombosis |
11 |
9,7% |
12,4% |
0,82 [0,66, 1,02] NS |
high |
|
Episodes of pulmonary embolism |
9 |
0,20% |
0,53% |
0,44 [0,18, 1,05] NS |
High |
|
Minor bleeding episodes |
9 |
9,9% |
10,7% |
0,91 [0,73, 1,13] NS |
High |
|
Major bleeding episodes |
8 |
1,9% |
2,1% |
0,89 [0,55, 1,43] NS |
High |
|
Death |
6 |
2,8% |
3,4% |
0,87 [0,66, 1,15] NS |
High |
The Meta-analysis showed no statistically significant differences between LMWH and UFH in all the assessed endpoints. A trend was identified, suggesting a higher clinical efficacy of low molecular weight heparins.
Discussion
The increasing prevalence of risk factors for venous thromboembolism, as well as the progress in diagnostic methods, leads to an increasing number of diagnosed cases. Along with an elevated risk of disease, the sales rates of low molecular weight heparins are steadily rising. In some countries, the costs of low molecular weight heparins is among the highest of all reimbursed drugs.
This analysis attempts to complement the studies, forming a base for consideration of the rationality of the use of low molecular weight heparins. The results of the meta-analyses enable a more accurate assessment of the clinical effectiveness of low molecular weight heparins, in comparison to individual studies. The results confirm the effectiveness and safety of LMWH in prevention of venous thromboembolism, while drawing attention to the fact that most of the assessed endpoints did not achieve statistically significant difference, compared to cheaper treatments, such as the physical methods or unfractionated heparin. This fact should be taken into consideration in the conditions, where cheaper therapies (as the above-mentioned UFH and the physical methods) are readily available.
The use of LMWH in the prevention and treatment of venous thromboembolism, when compared with unfractionated heparin, is more convenient in practice. It does not require the activated partial thromboplastin time (APTT) to be determined nor the use of infusion pumps. The easy use of LMWH, combined with their pharmacokinetic properties, allows for administration of the drug in outpatient settings or even at home. There is also a financial aspect of hospitalization, which has been omitted in this analysis. The most advantageous feature of low molecular weight heparins, compared to heparin, is the predictable relationship between dose and effect of anticoagulant, which translates into a dosage based on the weight of the patient, without laboratory monitoring.
The most common and also the most feared complication of both unfractionated heparin and low molecular weight heparins is bleeding. The risk of bleeding is higher in case of unfractionated heparin, however, the difference in the reported study was not statistically significant.
The present analysis leads to a surprising conclusion that the physical methods are highly effective, when compared to LMWH. Trials assessing the end point of deep vein thrombosis risk, included studies with ambiguous results. On the other hand, the meta-analysis did not confirm statistically significant superiority of LMWH. In case of a high probability of complications, including bleeding, and of the coexistence of additional risk factors, the use of physical methods is recommended as an effective thromboprophylaxis. They can be an alternative when contraindications to anticoagulants exist.
In the analyzed studies, there were no other significant, treatment-associated, adverse effects, other than bleeding incidents. This demonstrates an acceptable safety profile of low molecular weight heparins, compared to placebo [3-6], and a significantly better safety profile, compared to unfractionated heparin [7-15]. Significant clinical benefits, arising from their use, outweigh the potential risk of adverse effects, such as bleeding.
Conclusions
The results of this analysis demonstrate the effectiveness of low molecular weight heparins and safety of their use in prevention of deep vein thrombosis and pulmonary embolism. However, they also draw attention to the lack of statistical significance in a number of parameters versus other, less expensive methods, such as unfractionated heparin or physical methods (foot pump). Additionally, it should be noted that all the results of the meta-analyses take into account the realities of presented clinical trials and cannot be directly transferred into the reality of the Polish medical practice.
- Warwick D., Harrison J., Whitehouse S., Mitchelmore A., Thornton M. A randomised comparison of a foot pump and low-molecular-weight heparin in the prevention of deep-vein thrombosis after total knee replacement. J Bone Joint Surg Br. 2002 Apr;84(3):344 – 50
- Warwick D., Harrison J., Glew D., Mitchelmore A., Peters TJ., Donovan J. Comparison of the use of a foot pump with the use of low-molecular-weight heparin for the prevention of deep-vein thrombosis after total hip replacement. A prospective, randomized trial. J Bone Joint Surg Am. 1998 Aug; 80(8):1158–66
- Norgren L., Toksvig-Larsen S., Magyar G., Lindstrand A., Albrechtsson U. Prevention of deep vein thrombosis in knee arthroplasty: preliminary results from a randomised controlled study of lowmolecular weight heparin vs foot pump compression. Int Angiol1998; 17:93–6
- Blanchard J., Meuwly JY., Leyvraz PF., et al. Prevention of deep vein thrombosis after total knee replacement: randomised comparison between a low-molecular-weight heparin (nadroparin) and mechanical prophylaxis with a foot-pump system. J Bone Joint Surg [Br] 1999; 81-B:654–9
- Verso M. et al. Enoxaparin for the prevention of venous thromboembolism associated with central vein catheter: a double-blind, placebo-controlled, randomized study in cancer patients. J Clin Oncol. 2005 Jun 20; 23(18): 4057-62. Epub 2005 Mar 14
- Leizorovicz A. et al. Randomized, placebo-controlled trial of dalteparin for the prevention of venous thromboembolism in acutely ill medical patients. Circulation. 2004 Aug 17; 110(7): 874–9. Epub 2004 Aug 2
- Fraisse F. et al. Nadroparin in the prevention of deep vein thrombosis in acute decompensated COPD. The Association of Non-University Affiliated Intensive Care Specialist Physicians of France. Am J Respir Crit Care Med. 2000 Apr; 161(4 Pt 1): 1109–14
- Leclerc J.R. et al. Prevention of deep vein thrombosis after major knee surgery-a randomized, double-blind trial comparing a low molecular weight heparin fragment (enoxaparin) to placebo. Thromb Haemost. 1992 Apr 2; 67(4): 417–23
- Sherman D.G. et al. The efficacy and safety of enoxaparin versus unfractionated heparin for the prevention of venous thromboembolism after acute ischaemic stroke (PREVAIL Study): an open-label randomised comparison. Lancet. 2007 Apr 21; 369(9570): 1347–55
- Hillbom M. et al. Enoxaparin vs heparin for prevention of deep-vein thrombosis in acute ischaemic stroke: a randomized, double-blind study. Acta Neurol Scand 2002; 106:84–92
- Kleber F.X. et al. Randomized comparison of enoxaparin with unfractionated heparin for the prevention of venous thromboembolism in medical patients with heart failure or severe respiratory disease. THE-PRINCE Study Group. American Heart Journal 2003; 145 (4): 614–21
- Bergmann J.F. e al. A multicenter randomized double – blind study of enoxaparin compared with unfractioned heparin in prevention of venous thromboembolic disease in elderly in-patients bedridden for ana acute medical illness. Verlagsgesellschaft mbH 1996; 76 (4): 529–34
- Nurmohamed M.T. et al. A comparative trial of a low molecular weight heparin (enoxaparin) versus standard heparin for the prophylaxis of postoperative deep vein thrombosis in general surgery. Am J Surg. 1995 Jun; 169(6): 567–71
- McLeod R.S. et al. Subcutaneous Heparin Versus Low Molecular Weight Heparin as Thromboprophylaxis in Patients Undergoing Colorectal Surgery. ANNALS OF SURGERY Vol. 233, No. 3, 438–444
- Colwell C.W. Jr. et al. Efficacy and safety of enoxaparin versus unfractionated heparin for prevention of deep venous thrombosis after elective knee arthroplasty. Enoxaparin Clinical Trial Group. Clin Orthop Relat Res. 1995 Dec; (321): 19–27
- Colwell C.W. Jr et al. Use of enoxaparin, a low-molecular-weight heparin, and unfractionated heparin for the prevention of deep venous thrombosis after elective hip replacement. A clinical trial comparing efficacy and safety. Enoxaparin Clinical Trial Group. J Bone Joint Surg Am. 1994 Jan; 76(1): 3–14
- Fauno P. et al. Prophylaxis for the prevention of venous thromboembolism after total knee arthroplasty. A comparison between unfractionated and low-molecular-weight heparin. J Bone Joint Surg Am. 1994 Dec; 76(12): 1814–8
- Levine M.N. et al. Prevention of deep vein thrombosis after elective hip surgery. A randomized trial comparing low molecular weight heparin with standard unfractionated heparin. Ann Intern Med. 1991 Apr 1; 114(7): 545–51
- Avikainen V. et al. Low molecular weight heparin (enoxaparin) compare with unfractioned heparin in prophylaxis od deep venous thrombosis and pulmonary embolism in patients undergoing hip replacement. Annales Chirurgiae et Gynaecologiae 1995; 84: 85 – 90
- Senaran H. et al. Enoxaparin and heparin comparison of deep vein thrombosis prophylaxis in total hip replacement patients. Arch Orthop Trauma Surg (2006); 126: 1–5
- Spinal Cord Injury Thromboprophylaxis Investigators. Prevention of venous thromboembolism in the acute treatment phase after spinal cord injury: a randomized, multicenter trial comparing low-dose heparin plus intermittent pneumatic compression with enoxaparin. J Trauma. 2003 Jun;54(6):1116–24; discussion 1125–6Studies not available and not included:
- Bernstein R.A. Prevention of venous thromboembolism in patients with stroke – enoxaparin vs unfractionated heparin. Nat Clin Pract Neurol. 2007 Sep 11; [Epub ahead of print]Studies withdrawn as not meeting inclusion criteria:
- Ramos J. et al. Interventions for preventing venous thromboembolism in adults undergoing knee athroscopy. Cochrane Database of Systematic Reviews 2007; Issue 2. Art. No.: CD005259
- Handoll HHG. et al. Heparin, low molecular weight heparin and physical methods for preventing deep vein thrombosis and pulmonary embolism following surgery for hip fractures. Cochrane Database of Systematic Reviews 2002, Issue 4. Art. No.: CD000305
- Mismetti P. et al. Prevention of venous thromboembolism in internal medicine with unfractionated or low-molecular-weight heparins: a meta-analysis of randomised clinical trials. Thromb Haemost 2000; 83: 14–9
- Dentali F. et al. Meta-analysis: anticoagulant prophylaxis to prevent symptomatic venous thromboembolism in hospitalized medical patients. Ann Intern Med. 2007 Feb 20; 146(4): 278–88
- Vaitkus P.T. et al. Rationale and design of a clinical trial of a low-molecular-weight heparin in preventing clinically important venous thromboembolism in medical patients: the prospective evaluation of dalteparin efficacy for prevention of venous thromboembolism in immobilized patients trial. Vasc Med. 2002; 7(4): 269–73
- Spinal Cord Injury Thromboprophylaxis Investigators. Prevention of venous thromboembolism in the rehabilitation phase after spinal cord injury: prophylaxis with low-dose heparin or enoxaparin. J Trauma. 2003 Jun; 54(6): 1111–5
- Kearon C. et al. Comparison of fixed-dose weight-adjusted unfractionated heparin and low-molecular-weight heparin for acute treatment of venous thromboembolism. JAMA. 2006 Aug 23; 296(8): 935–42
- Marsland T. et al. Is once- or twice-a-day enoxaparin as effective as unfractionated heparin for the treatment of venous thromboembolism (VTE)? J Fam Pract. 2001 May; 50(5): 396
- Merli G. et al. Subcutaneous enoxaparin once or twice daily compared with intravenous unfractionated heparin for treatment of venous thromboembolic disease. Ann Intern Med. 2001 Feb 6; 134(3): 191–202
- Chong B.H. et al. Once-daily enoxaparin in the outpatient setting versus unfractionated heparin in hospital for the treatment of symptomatic deep-vein thrombosis. J Thromb Thrombolysis. 2005 Jun; 19(3): 173–8
- Ramacciotti E. et al. An open-label, comparative study of the efficacy and safety of once-daily dose of enoxaparin versus unfractionated heparin in the treatment of proximal lower limb deep-vein thrombosis. Thromb Res. 2004; 114(3): 149–53
- Levine M. et al. A comparison of low-molecular-weight heparin administered primarily at home with unfractionated heparin administered in the hospital for proximal deep-vein thrombosis. N Engl J Med. 1996 Mar 14; 334(11): 677–8
- Monreal M. et al. Comparison of subcutaneous unfractionated heparin with a low molecular weight heparin (Fragmin) in patients with venous thromboembolism and contraindications to coumarin. Thromb Haemost. 1994 Jan; 71(1): 7–11
- Lindmarker P. et al. Comparison of once-daily subcutaneous Fragmin with continuous intravenous unfractionated heparin in the treatment of deep vein thrombosis. Thromb Haemost. 1994 Aug; 72(2): 186–90
- Koopman M.M. et al. Treatment of venous thrombosis with intravenous unfractionated heparin administered in the hospital as compared with subcutaneous low-molecular-weight heparin administered at home. The Tasman Study Group. N Engl J Med. 1996 Mar 14; 334(11): 682–7
- Findik S. et al. Low-molecular-weight heparin versus unfractionated heparin in the treatment of patients with acute pulmonary thromboembolism. Respiration. 2002;69(5):440–4.
- Pérez de Llano L.A. et al. Multicenter, prospective study comparing enoxaparin with unfractionated heparin in the treatment of submassive pulmonary thromboembolism. Arch Bronconeumol. 2003 Aug; 39(8): 341–5
- Pini M. et al. Low molecular weight heparin (Alfa LHWH) compared with unfractionated heparin in prevention of deep-vein thrombosis after hip fractures. International Angiology 1989; 8: 134–9
- Quinlan D.J. et al. Low-molecular-weight heparin compared with intravenous unfractionated heparin for treatment of pulmonary embolism: a meta-analysis of randomized, controlled trials. Ann Intern Med. 2004 Feb 3; 140(3): 175–83
- Wirth T. et al. Prevention of venous thromboembolism after knee arthroscopy with low-molecular weight heparin (Reviparin): Results of a randomized controlled trial. Arthroscopy 2001;17(4):393–9.
- Marlovits S. et al. A prospective, randomized, placebo-controlled study of extended-duration post-discharge thromboprophylaxis with enoxaparin following arthroscopic reconstruction of the anterior cruciate ligament. Blood 2004;104 (11 Part 1): Abstract 1764
- Geerts W. et al. Thromboprophylaxis after major trauma – a double-blind RCT comparing low dose Heparin (LDH) and the low molecular weight heparin (LMWH), Enoxaparin [Abstract]. Journal of Trauma 1995; 39(1): 159
- Grosset A.B. et al. Enoxaparin, a low-molecular-weight heparin suppresses prothrombin activation more effectively than unfractionated heparin in patients treated for venous thromboembolism. Thrombosis research 1997; 86 (5): 349–54
- Diener H.C. et al, for the PROTECT Trial Group. Prophylaxis of thrombotic and embolic events in acute ischemic stroke with the low-molecular-weight heparin certoparin: results of the PROTECT Trial. Stroke 2006; 37: 139–44Studies used in introduction:
- Chęciński P. et al. Żylna choroba zakrzepowo-zatorowa. Przew Lek 2006; 5: 54–69
- Filipecki S. Epidemiologia i czynniki ryzyka żylnej choroby zakrzepowo-zatorowej. Warszawa 2002; 4-10