A cost-effectiveness analysis of programmable baclofen pump therapy in children with spastic cerebral palsy
-
Copyright
© 2012 PRO MEDICINA Foundation, Published by PRO MEDICINA Foundation
User License
The journal provides published content under the terms of the Creative Commons 4.0 Attribution-International Non-Commercial Use (CC BY-NC 4.0) license.
Authors
| Name | Affiliation | |
|---|---|---|
Jacek Walczak |
Arcana Institute, Płk S. Dąbka 8, PL 30-732 Kraków, Poland |
|
Marek Jóźwiak |
The Children\\\'s Orthopaedic Surgery and Traumatology Clinic, University of Medical Sciences in Poznań, Fredry 10, PL 61-701 Poznań, Poland |
|
Anna Jasińska |
Arcana Institute, Płk S. Dąbka 8, PL 30-732 Kraków, Poland |
|
Magdalena Garbacka |
Arcana Institute, Płk S. Dąbka 8, PL 30-732 Kraków, Poland |
|
Grzegorz Obrzut |
Arcana Institute, Płk S. Dąbka 8, PL 30-732 Kraków, Poland |
|
Wiktor Dardziński |
Arcana Institute, Płk S. Dąbka 8, PL 30-732 Kraków, Poland |
Methods: The cost-utility analysis of intrathecal baclofen treatment was performed from the perspective of public payer in 6 years' time horizon, using Markov model, constructed in the frame of computer program TreeAge® Pro Suite 2008. The target population are children with severe spasticity in cerebral palsy which is refractory to current treatment options. Annual discount rate was set equal to 5% both for costs and the health effects. Assumptions of the analysis were tested in one- and multiway sensitivity analysis.
Results: A gain of one quality adjusted life year (QALY) in child with spasticity costs 60 224 PLN (17 126 €) when standard treatment is replaced with baclofen pump therapy. Conclusion. Intrathecal baclofen pump therapy in children with spastic cerebral palsy is a cost-effective strategy in Poland.
Introduction
Cerebral palsy is a non-progressive but non-constant disorder of posture and motion in children caused by central nervous system damage during an early stage of development, coexisting with some other symptoms: cognitive functions and speech development disorders, epilepsy, mental impairment, sight and hearing disorders. In Poland there are 2-3 children diagnosed with cerebral palsy per 1000 live births, giving 20-25 thousand children with this disease.
The purpose of spasticity management is to improve motor activity, preventing complications, decreasing pain and facilitation of nursing. Currently, there are several ways of increased muscular tone treatment: rehabilitation, farmacotherapy with oral spasmolytic medicines, focal treatment with botulinum toxin and orthopaedic surgeries aiming to reduce results of muscle contractures. The therapy with botulinum toxin in cerebral palsy in Poland is financed by NHF (National Health Fund) as the therapeutic program of spasticity treatment. Small invasiveness and lack of side-effects are advantages of rehabilitation, but on the other hand the effect of reducing spasticity is short-lasting and temporary. One of the recent methods is a therapy based on subarachnoidal infusion of baclofen by the means of a programmable baclofen pump. This treatment is recommended especially for patients who suffer from severe, chronic spasticity, when conventional pharmacological therapy is ineffective in their case and when the outer pump with a transcutaneous catheter or the injections of a medicine directly into the spinal cord or into an internal organ are either not effective enough or uncomfortable for a patient.
In the analysis the costs and the health benefits of treatment with a programmable baclofen pump and the currently financed therapy of spasticity in children were compared.
Materials and methods
Population
The target population are children with significant, chronic spasticity of cerebral origin, who are refractory to available treatment methods and showing positive response to baclofen test dose.
Intervention
Intrathecal baclofen (ITB) continuously administered by an implantable, programmable infusion pump was assessed. Concomitant therapy includes rehabilitation, other medicines and orthopaedic surgery. Baclofen administered intrathecally in a continuous way by an infusion pump is recommended for treatment of patients with severe spasticity that cannot be effectively managed with oral baclofen.
Comparator
The compared strategy is current standard therapy, which includes rehabilitation, oral pharmacotherapy, focal treatment with botulinum toxin and orthopaedic surgery.
Clinical outcomes
The health effects in the cost-utility analysis are measured by the quality adjusted life-years (QALY) gained.
Perspective
The cost-utility analysis was performed from the perspective of public payer for health services in Poland (National Health Fund).
Time horizon
For battery working time is limited, the process of pump implantation must be repeated every 5-7 years [3,4]. When the battery runs down the whole pump has to be replaced with a new one. Pump lifespan is also partially determined by durability of the membrane, through which the insertions supplementing medicine are made. The time horizon of the cost-utility analysis was 6 years, assuming that during such a period one infusion pump is used.
Analytical technique
In order to compare the two treatment options a cost-utility analysis (CUA) was carried out, the results of which are the additional costs for the quality adjusted life-year (QALY) gained when replacing standard treatment with ITB therapy. The cost-utility analysis was performed using Markov model, constructed in the frame of computer program TreeAge® Pro Suite 2008. Annual discount rate was set equal to 5% both for costs and the health effects. Assumptions of the analysis were tested in one- and multiway sensitivity analysis.
Data resources
The resources of data concerning efficacy and safety of the two treatments compared. Resource use were clinical trials found through systematic review method of retrieval, other economic analyses and opinions of medical experts.
Clinical effectiveness based on systematic review
The aim of the systematic review [5] was to evaluate the clinical effectiveness of the intrathecal baclofen in the treatment of spasticity due to various etiology on the basis of systematic literature review.
Only randomized trials including a control group were included into the analysis. One study met the inclusion criteria (randomized controlled trial, comparing intrathecal baclofen therapy plus conventional therapy versus conventional therapy alone), conducted in patients with intractable spastic cerebral palsy (CP). No randomized studies regarding clinical efficacy of baclofen pumps in spasticity treatment in other disease entities (multiple sclerosis, stroke, brain injury, spinal cord injury) were found. Thus, analysis for these indications was based on non-randomized trials without control groups.
The comparative analysis in patients with CP revealed a positive impact of continuous intrathecal baclofen infusion (CITB) measured in Visual Analogue Score (VAS) after 6 and 12 months and in Child Health Questionnaire – Parent Form 50 (CHQ-PF50) after 12 months of therapy. The safety analysis in an open label phase revealed 51 cases of adverse events (AEs) and 29 device adverse events (DAEs). The results suggests high clinical efficacy and safety of the infusion of intrathecal baclofen in the therapy of spasticity of CP.
Model description
The decision model was performed using Markov model to compared ITB and standard therapy in 6 years’ time horizon, whereas the length of a single cycle (frequency of patient’s state changes) was set to be 1 year. In the constructed model the states representing crucial events in the course of cure of spasticity from clinical and economical points of view were singled out. In any fixed cycle a patient is only in one state and a transition to another state and staying within the same state happen with prescribed probabilities. There are costs, utilities and transition probabilities attributed to each state and this allows to evaluate long-term costs and health effects of the therapies analyzed. A graphical depiction of the model structure (health states and possibilities of transitions between them) is displayed on Figure 1.
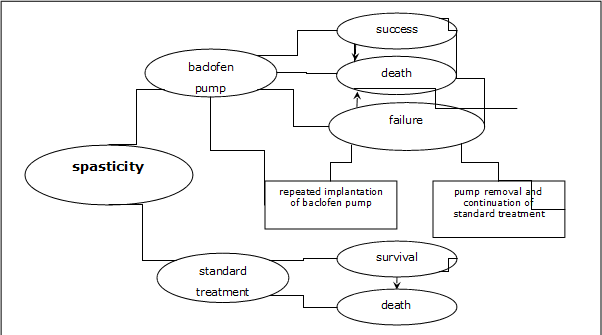
In the compared strategies of spasticity treatment the following health states were singled out:
baclofen pump:
- success,
- failure,
- death.
standard treatment:
- survival,
- death.
The state of "success" in case of baclofen pump therapy was defined as an improvement of health condition, meaning the increased mobility, self-care and functional status, improvement of comfort and sense of well-being and decrease in pain. This state has ascribed costs connected with ITB and concomitant therapies and corresponding utility value. The model takes into consideration a possibility of the therapy failure, too, because of adverse events or lack of effectiveness (the state of "failure"), which can lead to the necessity of baclofen pump removal. In case of therapy withdrawal (because of severe side-effects making intrathecal infusion of baclofen impossible or the lack of getting the expected health effect) a patient switches to the standard treatment, but in some instances the baclofen pump can be implanted again allowing the intrathecal baclofen therapy to be continued. If the therapy fails then there are additional costs of removal of the baclofen pump and possibly the costs of repeated implantation; furthermore one takes into account a reduction in health state utility value.
The target population are children, who are refractory to available treatment methods, so in the standard therapy there are the state of "survival" involves average annual costs per patient with spasticity and an appropriate value of the utility index. All the states in the model take into consideration treatment costs of possible adverse effects/medical incidents connected with the therapies applied. With the same probability one can pass from each state to the state "death" (based on the evaluated death risk in cerebral palsy).
Cost analysis
In order to calculate the costs of both therapies compared the perspective of public payer (National Health Fund) was assumed. The following sources of direct costs linked to the medical treatment of spasticity were taken into consideration:
- ITB treatment: cost of baclofen test, cost of pump implantation/removal, costs of pump refill visits, costs of complications connected with ITB therapy and costs of concomitant therapy (rehabilitation, other medicines and surgical treatment),
- standard treatment: costs of rehabilitation (including orthopaedic equipment and outpatient visits), oral pharmacotherapy, focal treatment and surgical operations, costs of standard therapy (oral medicines and botulinum toxin) complications and costs of curing pressure sores.
An exact evaluation of expenditures resulting from spasticity treatment in Poland was made according to experts’ estimations (questionnaires sent to specialist centers of spasticity treatment and rehabilitation in Poland) and available literature concerning the topic. The pricing of the discussed resources was carried out on the base of the National Health Fund charges (concerning medical services and medicines by the new clearing system based on Diagnosis Related Groups classification, valid since July 1, 2008) in agreement with perspective of the analysis. Evaluated average annual costs connected with spasticity management per one patient with baclofen pump and cured with standard treatment is displayed on the Table 1.
Table 1. Costs of spasticity treatment – ITB therapy and standard treatment
| Component of costs | Child with baclofen pump [PLN] | Child cured with standard treatment [PLN] | |
| Baclofen test | 624 | - | |
| Implantation/reimplantation of baclofen pump * | 33 700 | - | |
| Pump removal procedure | 7 700 | - | |
| Control visits in order to refill pump | The 1st year | 11 060 | - |
| Consecutive years | 12 640 | - | |
| Complications resulting from ITB treatment | At most 60 days after pump implantation | 276,20 | - |
| At least 60 days after pump implantation | 511,63 | - | |
| Standard treatment | Rehabilitation | 5 293 | 7 439 |
| Orthopaedic equipment | 6 009 | 6 009 | |
| Oral medicines | 8,2 | 58,1 | |
| Focal treatment with botulinum toxin | 287,1 | 2 647,6 | |
| Orthopaedic surgery | 1 285 | 3 597,9 | |
| Outpatient visits in an orthopaedic clinic | 109,5 | 109,5 | |
| Complications resulting from standard therapy | Oral medicines | - | 7,96 |
| Botulinum toxin | - | 414 | |
| Pressure sores treatment | - | 154 | |
*including cost of procedure, cost of medical device and Lioresal Intrathecal, cost of in-patient stay
Considering the 1-year cycles in the cost-utility model, the costs of ITB complications treatment were counted in the year of therapy. The costs of complications arising from the ITB treatment were calculated as equal initially to 276 PLN and 48 PLN in the successive years of the time horizon of analysis (years 1-6).
Parameters of the model
Population
It was supposed in the model that patient beginning therapy is 12 years old on average (mean age was evaluated on the basis of patients’ age data from clinical trials concerning effectiveness of the baclofen pump treatment).
Utilities
Utility weights for various health states occurring in the model com from the analysis of de Lissovoy et al. [6]. The lowest average utility value was obtained when there was no ITB therapy (in the population whose spasticity was not controlled by standard therapy) and it equals 0,125 (SD=0,058). The utility increased slightly in the initial phase of ITB therapy (with a pump) to the value of 0,162 (SD=0,048). As a result of the long-term treatment the average utility value attained 0,401 (SD=0,106) [6].
Withdrawal from therapy
Depending on a trial and period of time, the percentage of patients who withdraw from ITB therapy fluctuates from 4% [7,8] up to about 48% [9,10]. Probability of quitting the therapy because of adverse effects or lack of effectiveness in a 1 year cycle of treatment in the model was determined on the basis of the trial of Vender 2006 [11] since there were long period of time and a numerous group of patients considered and additional information about the number of patients with a repeated pump implantation procedures. The obtained percentage of patients withdrawing from the therapy (Pwithdraw=22,41%) was transformed into a risk of withdrawal per one cycle of the model (pc=4,95), assuming equal probability of therapy withdrawal in each cycle and using the formula (pc=1-(1-Pwithdraw)1/c), where c denotes the number of years in the trial. The probability of a repeated pump implantation after its removal was evaluated according to the percentage of total abandoning of therapy also from Vender et al [11] (it is equal to 0,885).
In case that severe unwished effects have shown up, which result in compulsory removal of the baclofen pump, the health state utility index was assumed to lower by 50% for a half of year.
Mortality
Information on mortality rate of children with cerebral palsy was calculated on the basis of general mortality of children population with cerebral palsy in United Kingdom excerpted from Hemming 2005 [12]. No analogical Polish data were identified in the search. The probability of 5-years’ survival for a 10-years old child was evaluated as 0,973 and as 0,963 for a 15-years old child.
Annual probability of death for patients in separate age groups was calculated using the formula (pdeath=1-exp(-h)), where "h" denotes the relative hazard, it equals 0,00553 for children between 10 and 15 years old and 0,00756 for those between 15 and 20 years old.
Results
Cost-utility analysis
A compilation of health effects and costs per one patient, which were obtained as a result of the cost-utility model calculation is displayed in Table 2.
Table 2. Compilation of costs and effects for intrathecal baclofen and standard therapy of spastic cerebral palsy
| Parameter | Baclofen pump | Standard therapy |
| 1st year of therapy [PLN] | ||
| Treatment costs | 60 367 | 19 637 |
| Costs in 6 years’ time horizon [PLN] | ||
| Treatment costs | 142 369 | 64 252 |
| Including: | ||
| Treatment costs connected with ITB pump (without concomitant treatment) | 105 535 | - |
| Concomitant treatment costs | 36 834 | - |
| Pump removal and possible repeated implantation costs | 7 873 | - |
| Rehabilitation costs | 27 247 | 38 051 |
| Costs of adverse events treatment (inc. cost of pump removal) |
773 | 1 449 |
| Health outcomes | ||
| Quality adjusted life-years (QALYs) in a 1-year time horizon | 0,319 | 0,122 |
| Quality adjusted life-years (QALYs) in a 6-years time horizon) | 1,936 | 0,639 |
The incremental cost-utility ratio (ICUR) for the comparison of continuously administered ITB therapy to standard therapy (ST) of spasticity in a 6 years’ time horizon was determined from the following formula:
ICUR = (costITB - CostST) / (EffectITB - EffectST)
The cost-utility analysis proved that ITB treatment together with concomitant therapy is more expensive but more effective than standard therapy. Total costs of baclofen pump therapy amount to 142 369 PLN (40 485 €)1, whereas the costs of standard therapy come to 64 252 PLN (18 271 €) in 6 years’ time horizon. The incremental cost-utility ratio (ICUR) equals 60 224 PLN/QALY (17 126 €/QALY). In conformity with the recommendation of the AOTM2 Consultative Council [13] and a principle accepted by WHO (World Health Organization) and on the basis of CSO3 [14] data the profitability threshold in Polish circumstances is 91 914 PLN (26 137 €) for a unit of effect. Thus, the ITB therapy is a cost-effective strategy in Poland.
Sensitivity analysis
In order to investigate an influence of a change of the key parameters and the settings of model on results of the cost-utility analysis, one-way and multi-way sensitivity analyses were performed. The results of one-way sensitivity analysis confirmed that ITB therapy of spasticity in children remains more expensive, but still more effective than standard treatment (Table 3). The value of ICUR fluctuated within -40,7% (assuming that the frequency of visits in order to refill the baclofen pump is once per 3 months) up to +26,5% (assuming higher probability of abandoning the therapy because of adverse effects or lack of effectiveness of the therapy), only when 1 year’s time horizon was considered the ICUR ratio value jumped to 206 793 PLN (growth of 243% compared to the basic value). Changes of the remaining parameters have a minor influence on the result of analysis (maximal deviation is 19%). In each of the cases under consideration (except 1 year’s time horizon) in the sensitivity analysis, the ITB therapy remained a cost-effective technology. The multi-way sensitivity analysis demonstrated that the biggest changes of the ICUR from the basic value occurred when the minimal (maximal) utility value for baclofen pump therapy and simultaneously the maximal (minimal) utility value for standard therapy (changes respectively of +67,5% and -28,7%) are supposed.
Table 3. Results of one-way sensitivity analysis
| No. | Parameter | Notes | ICUR [PLN/QALY] |
| 1. | Basic value | 60 224 | |
| 2. | Increased cost of baclofen test | Diagnostic stay longer than 2 days | 61 630 |
| 3. | Frequency of visits in order to refill the pump – once per 3 months | 35 705 | |
| 4. | Minimal costs of rehabilitation | Based on questionnaires | 64 604 |
| 5. | Maximal costs of rehabilitation | 54 533 | |
| 6. | Minimal costs of focal treatment | 63 855 | |
| 7. | Maximal costs of focal treatment | 49 843 | |
| 8. | Complications of ITB treatment costs -30% | 60 103 | |
| 9. | Complications of ITB treatment costs +30% | 60 345 | |
| 10. | Lack of standard treatment complications | 61 323 | |
| 11. | No botulinum toxin treatment in baclofen pump treatment patients group | 59 110 | |
| 12. | No botulinum toxin treatment in standard treatment patients group | 71 564 | |
| 13. | Equal costs of orthopaedic treatment in both groups | 61 986 | |
| 14. | Costs of orthopaedic treatment -50% in both groups | 61 105 | |
| 15. | Costs of orthopaedic treatment +50% in both groups | 59 343 | |
| 16. | No necessity of orthopaedic surgeries in patients cured with baclofen pump therapy | 59 245 | |
| 17. | No necessity of orthopaedic surgeries in patients cured with standard therapy | 62 965 | |
| 18. | Lower probability of ITB pump therapy withdrawal | Based on Gilmartin 2000 | 56 876 |
| 19. | Higher probability of ITB pump therapy withdrawal | Based on Murphy 2002 | 76 180 |
| 20. | No costs and health effects discounting | 56 468 | |
| 21. | 1 year’s time horizon | 206 793 | |
| 22. | Minimal utility values | Based on de Lissovoy 2007 |
67 712 |
| 23. | Maximal utility values | 54 227 | |
| 24. | 5% discount rate for costs and 0% for health effects | 51 948 | |
| 25. | 5% discount rate for costs and 3,5% for health effects | 57 696 |
Discussion
No other economical analysis estimating profitability of using ITB therapy in spastic cerebral palsy in Poland were identified, hence an external validation was conducted on basis of analyses performed for other countries, and so for other health care systems. As a result of search of economical analyses (databases: PubMed, Cochrane Library, CRD Database, NICE) two analyses estimating the profitability of baclofen pump treatment of spasticity in children were found (Hoving 2008 [15], de Lissovoy 2007 [6]). In the analysis of Hoving 2008 standard therapy was defined to include physiotherapy, speech therapy, occupational therapy, while in the analysis of de Lissovoy 2007 [6] – as a conventional medical and surgical care. Present analysis in concordance with the currently used clinical practice in Poland, defines standard treatment of major spasticity as an entirety of activities serving to improve the functional conditions. The calculations were made for 6 years’ time horizon, whereas in both abovementioned foreign analyses the results were presented for shorter periods: 1 year – Hoving 2008 [15] and 5 years – de Lissovoy 2007 [6].
The analysis of de Lissovoy 2007 [6] seems to be the most similar with regard to the method applied (similar time horizon, health state utilities in the model were evaluated on the basis of the analysis of de Lissovoy [6]. Results presented in de Lissovoy [6] are consistent with the results of present analysis with respect to the health effects obtained. Using ITB therapy in comparison to standard therapy one gains 1,19 QALYs in a 5 years’ time horizon. In this analysis ITB therapy in 6 years’ time horizon together with concomitant therapy compared to standard therapy, allows to gain 1,30 QALYs.
The ICUR ratio obtained in the analysis of de Lissovoy [6] (42 000 $/QALY) does not exceed profitability threshold for a unit of effect in American conditions (it is a cost-effective technology). The difference of final ICUR result compared to present analysis may stem from different discount rate assumed in the analysis of de Lissovoy [6] (3% for costs and health effects), shorter time horizon (5 years) and, presumably, distinct cost data (there was no detailed information about it, only the total cost calculated on the basis of resource use in a group of a dozen or so of children; in both compared groups therapy costs were significantly higher than in Polish conditions). An additional difference between the discussed analyses is the approach to possible complications connected with ITB treatment. Merely, in the analysis of de Lissovoy [6] the treatment costs of adverse effects were not taken into account in basic variant (these costs were considered in an extra scenario, in which the final result was observed to reach 45 700 $/QALY). Moreover, patients’ withdrawal from baclofen pump therapy was not entertained, while in present analysis both the costs of adverse effects treatment (in both of compared groups) and costs of possible baclofen pump removal – and of repeated implantation in some circumstances – together with decrease of the health state utility which follows side-effects, were all considered. In the Dutch analysis of Hoving 2008 [15] effectiveness of baclofen pump therapy was measured in the VAS (Visual Analogue Score) scale, still the life quality was estimated using the EQ-5D questionnaire. In the analysis of Hoving 2008 gaining one additional QALY costs 32 737 € in 1 year’s time horizon. Authors of the analysis did not gave the results in a form of QALY and it complicates a comparison of the difference of health effects to those from the present analysis. The ICUR ratio obtained in the Dutch analysis does not surpass profitability threshold (80 000 €) for a unit of effect – the infusion pump therapy is a cost-effective technology.
1 1€=3,1566 PLN; mean echange rate in 2008
2 Polish Health Technology Assessment Agency
3 Polish Central Statistical Office












