Real-life treatment patterns and medication costs in patients with hypertension treated with ramipril monotherapy or ramipril loose and fixed combinations in Poland
-
Copyright
© 2015 PRO MEDICINA Foundation, Published by PRO MEDICINA Foundation
User License
The journal provides published content under the terms of the Creative Commons 4.0 Attribution-International Non-Commercial Use (CC BY-NC 4.0) license.
Authors
Background: Real-World Evidence (RWE) provides insights into patient outcomes reflecting current practices under existing healthcare system conditions. Longitudinal patient data are a source of information that can be used to track treatments for anonymised individual patients over time. Arterial hypertension is one of the most common chronic diseases in Poland, leading to serious complications, especially if undiagnosed and not properly controlled. Fixed-dose combinations of antihypertensive drugs become more and more popular in the treatment of hypertension. The objective of this study was to analyze real-life treatment patterns and medication costs in patients with hypertension treated with the most popular ACEI in Poland - ramipril, in monotherapy, loose and fixed combinations.
Methods: This analysis was based on the longitudinal prescription database (IMS “LRx”) of individual, anonymized patients structured by product, prescribing doctor specialty and patient age group and sex, sourced from dispensing transactions of 3367 chain and non-chain pharmacies, in the Polish open-care market. The period of analysis covers 20 months (January 2014 – August 2015). It is based on 1 488 053 patients and 9 023 582 prescriptions. Cost calculations were performed from payer’s and patient’s perspectives.
Results: Owing to the availability of multiple treatment options, patterns in hypertension treatment in Poland are very complex, even when considered only at the molecule level. Furthermore, switching to fixed-dose combination therapy based on ramipril resulted in savings for patients as well as the public payer.
Conclusion: Wider use of fixed-dose combinations is a cost saving option both for patients and the public payer.
Introduction
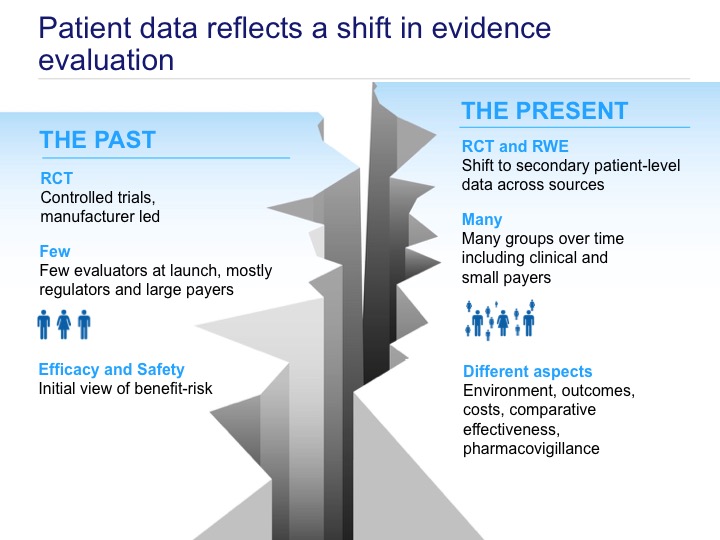
“The notion that evidence can be reliably placed in hierarchies is illusory (…). Observational studies too have defects but they also have merit. Decision makers need to assess and appraise all the available evidence” This statement made by Sir Michael Rawlins - the President of the National Institute for Health and Care Excellence - supports reaching beyond randomized controlled trials as a source of information on health technologies [1]. The term Real-World Evidence (RWE) or Real-World Data (RWD) comprises various types of data sets including information about epidemiology, effectiveness, safety, and costs of treatment, all generated and analyzed outside the framework of randomized controlled trials (RCTs) [2]. RWE provides insights into patient outcomes in a real-life setting, reflecting current practices under existing healthcare system conditions. It enriches information on efficacy obtained from clinical trials by adding data on effectiveness usually collected from a broader patient population (fig. 1).
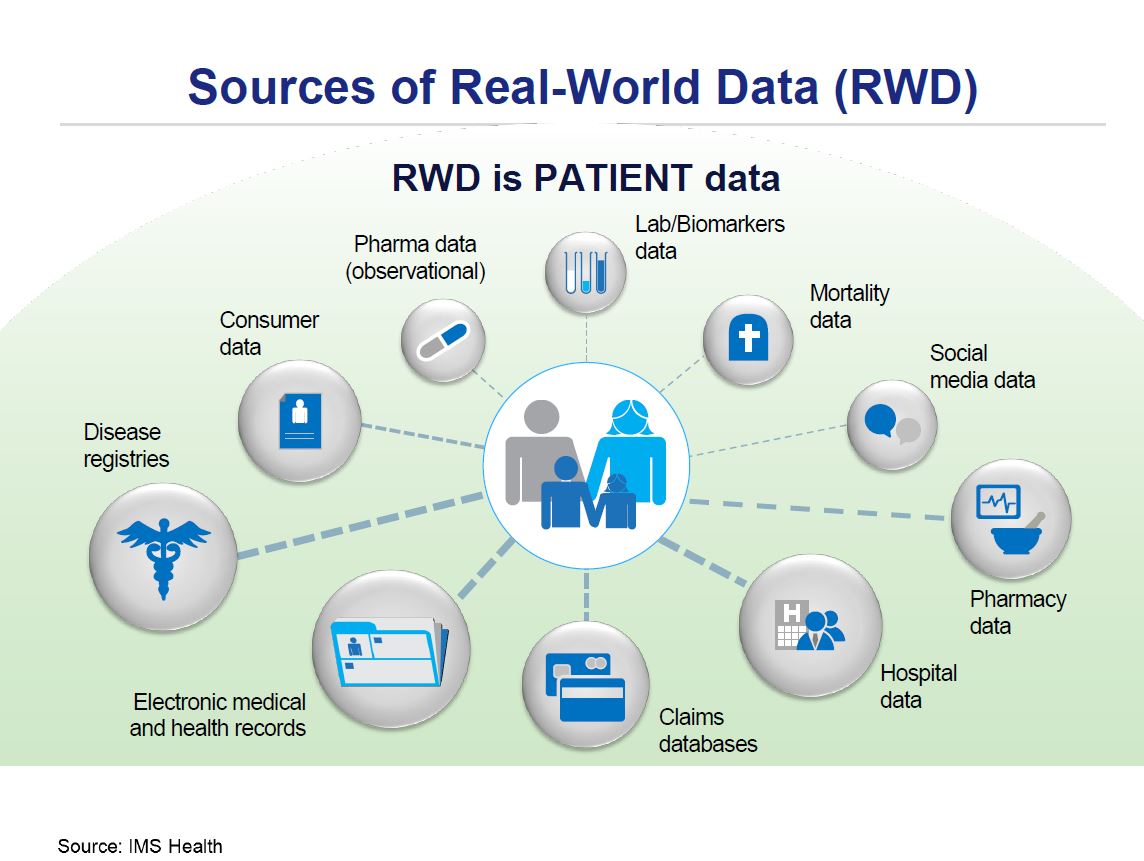
RWE comprises a wide scope of data sources: electronic medical records (EMRs), (payers/insurers) claims data, national surveys, disease specific registries, surveillance systems, online communities, data on prescription patterns, marketing, sales and distribution, (human) genome data. If we treat these groups as primary data sources, then secondary sources based on processed, analyzed and researched data can be further distinguished. Observational studies frequently use retrospective data from administrative databases or EMRs. In contrast, a cohort study is a longitudinal study where a group of persons with the same characteristics and defined exposure is followed in time until the outcome of interest occurs. Sometimes patient-reported outcomes (PROs) are measured: in this case information is provided by patients themselves (fig.2) [2].
Among the main stakeholders using RWE in medicine and healthcare one can list: regulatory agencies, (state) payers, (state) insurance companies, healthcare providers, health technology agencies, clinicians, patients, manufacturers, and consultants. In Poland, key stakeholders have not yet fully embraced RWE as an evidence source, despite the fact that their involvement seems to be crucial in the further development of this domain. The recently performed analysis based on an exploratory survey to map current awareness and expectations of physicians, related to practical outcomes data, showed significant interest and broad spectrum of possible actions to improve formal use of RWE in Poland [3]. Some of these efforts have already started: e.g. related to the health technology assessment (HTA) guidelines update currently performed by an expert group at the Polish Agency of Health Technology Assessment and Tariff System [4]. However, still few guidelines exist regulating usage of RWE in detail [2,5,6], and some of them are devoted to one type or one therapeutic area only [7,8].
Scientific quality and validity of information generated from RWE depend on sample size, representativeness vs. prevalence and incidence of the disease, completeness and clarity of the parameters coupled with the level of standardization [2].
Longitudinal patient data are generally considered as any anonymized patient- level information that can be tracked over time for an individual patient. In accordance with respective data privacy legislation, no personal data regarding patient, prescriber or pharmacy are collected or stored. As a result, such database contains exclusively anonymized information. Longitudinal patient information can help many stakeholders in the healthcare sector answer patient related questions, learn how medicine in real-world is practiced over time, along with actual treatment outcomes. This insight, coupled with cost data, can be useful in decision making processes leading to further optimized allocation of health care resources both at central, regional and individual practice levels[2].
Hypertension is one of the most common chronic diseases in Poland, leading to serious complications, especially if undiagnosed and not controlled properly. Ischemic heart disease, myocardial infarction and stroke are the leading causes of death worldwide and, despite a significant improvement [9], are still challenges for the healthcare sector in Poland. Diuretics, beta-blockers, angiotensin converting enzyme inhibitors (ACEI), angiotensin receptor antagonists and calcium antagonists are commonly used as a first-line therapy in arterial hypertension monotherapy. Unfortunately, monotherapy is often insufficient in controlling patients’ condition, requiring treatment with more than one antihypertensive agent. Therefore fixed-dose combinations of antihypertensive drugs become more and more popular in the treatment of arterial hypertension. They are also recommended by many guidelines, including Polish ones, as an effective and relatively safe therapeutic option [10]. Still the efficacy of cardiovascular prevention is hampered by several problems including: monitoring; inadequate choice of medication; poor compliance and adherence to treatment; and sometimes cost of treatment. For these reasons all main stakeholders, medical professionals, (public) payers and patients themselves should be interested in selecting effective and cost-effective treatment options.
The objective of this study was to analyze real-life treatment patterns and medication costs in patients with hypertension treated with the most popular ACEI in Poland - ramipril in monotherapy, loose and fixed combinations.
Methods
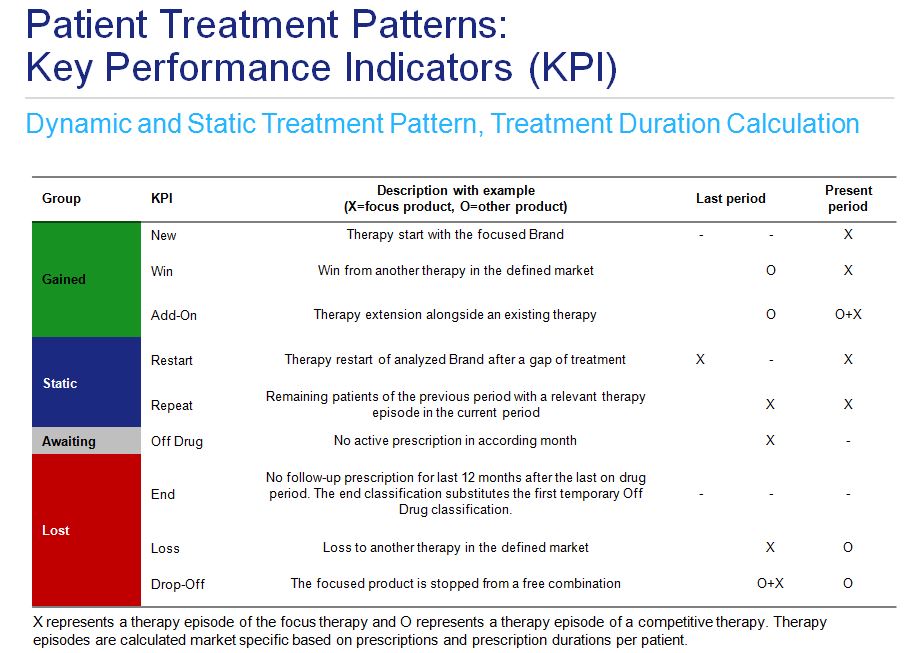
This analysis is based on longitudinal prescription database of individual anonymized patients (IMS “LRx”) structured by product, prescribing doctor specialty and patient age group and sex, sourced from dispensing transactions in the Polish open-care market, from 3367 chain and non-chain pharmacies (out of a total of 14 372 pharmacies in Poland in September 2015). All analytics are patient centric and market specific, calculated on an individual transaction basis collected every month from each pharmacy. Treatment dynamics considerations are based on a strict episode concept. Therapy episodes are calculated based on prescriptions and their durations. Free combinations of drugs in the same market caused by prescriptions on different days or by different doctors are taken into account in order to determine therapy combinations. If prescription duration is not available in the data, it is calculated based on existing information. Treatment pattern model was based on static and dynamic sub-models. The first one took into account initiation of treatment, treatment change and adding a new therapy to an existing one, as well as a permanent or temporary treatment termination. Continuation or restart of the therapy was considered in the static sub-model. (fig. 3).
Total volume and value of ramipril and ramipril combinations market was derived from the IMS Pharmascope database, and prices sourced from the drug prices database IMS Refundator, created from the extant reimbursement list issued by the Ministry of Health as of November 2015.
Cost calculations were performed from payer’s and patient’s perspectives.
This analysis covers the 20 months period from January 2014 – August 2015. It’s based on 1 488 053 patients and 9 023 582 prescriptions.
Results
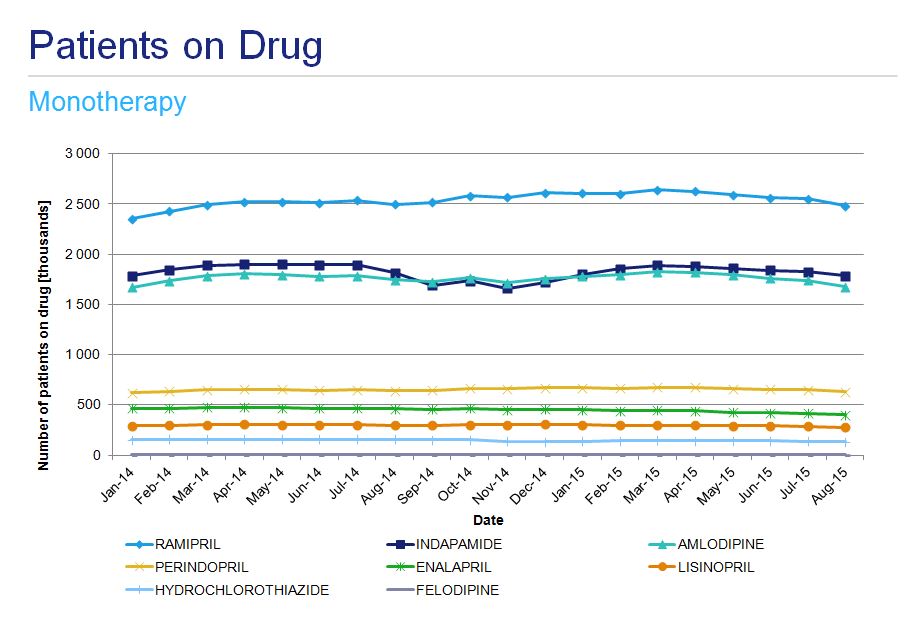
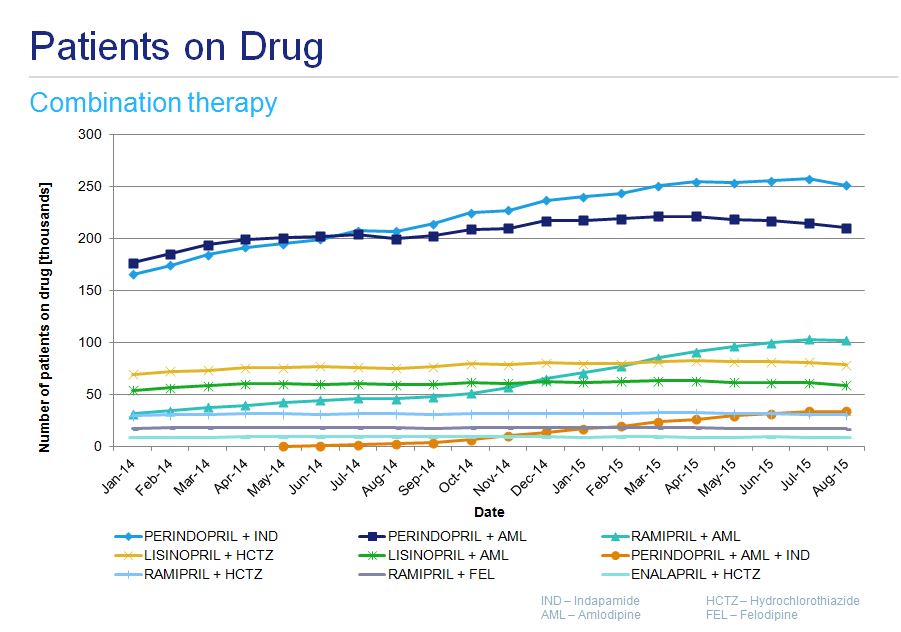
Ramipril is the most frequently prescribed angiotensin converting enzyme inhibitor (ACEI) in Poland with approximately 300 000 patients treated. Its popularity was the primary reason this drug was chosen for analyses. The second most popular drug - perindopril - is used by approximately 80 000 patients. From January 2014 to March 2015 the number of patients treated with ramipril grew month on month to reach a plateau, since when there has been a slow decline. In the 21 month period under analysis, the number of patients on perindopril was relatively stable. Antihypertensive drugs from different therapeutic classes, as well as those used in fixed dose combinations like indapamide, hydrochlorothiazide or felodipine, were also analyzed for the same period (fig. 4).
A number of patients treated with a fixed dose combinations is much lower with 30 000 and 25 000 patients receiving perindopril- indapamide and perindopril- amlodypine products, respectively. Ramipril- amlodipine combination, although not the most commonly prescribed, is the most dynamically increasing group of products (fig. 5).
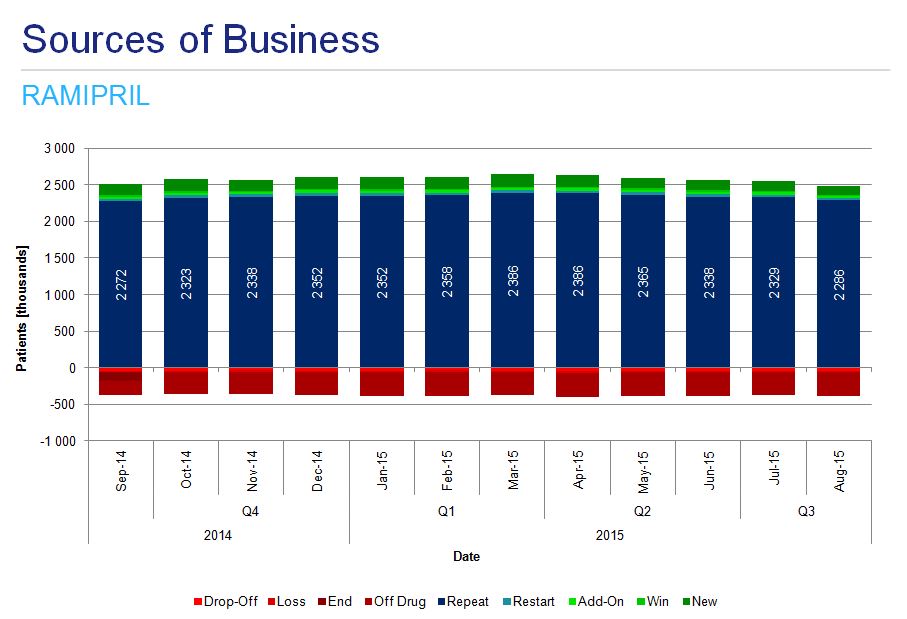
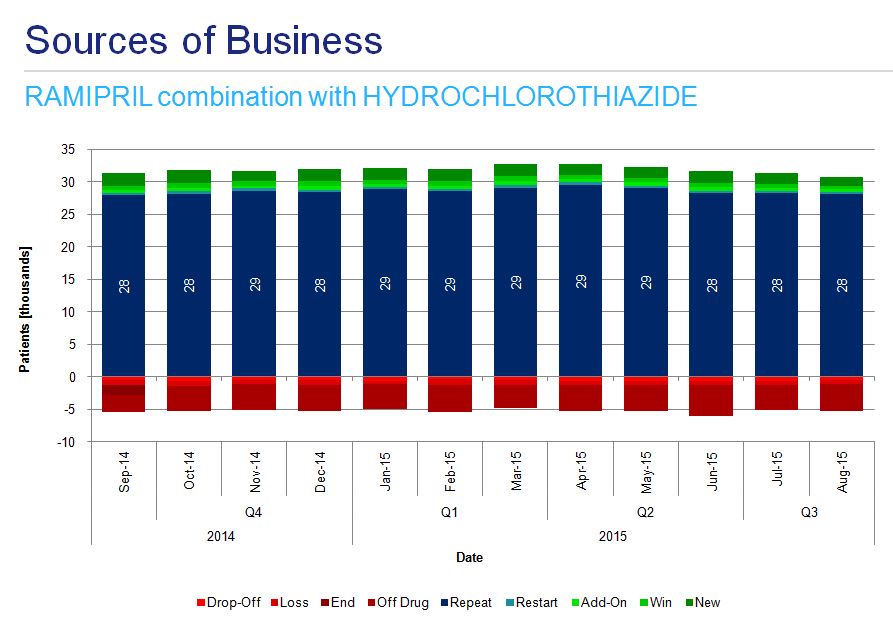
A detailed analysis of ramipril monotherapy treatment patterns shows the majority of patients repeat the treatment, there are a substantial number of off-drug patients in each period and there are many newly initiated patients (fig. 6)
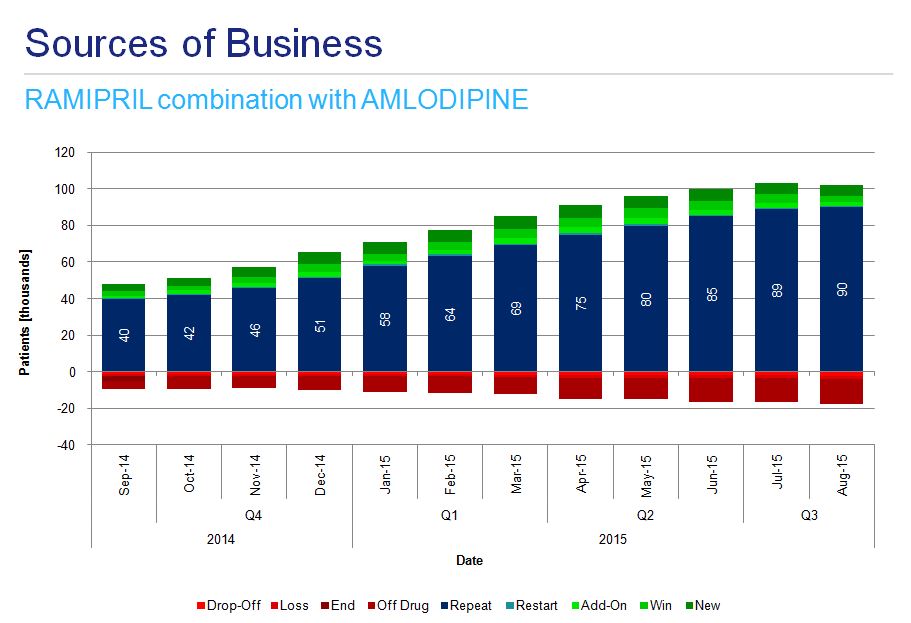
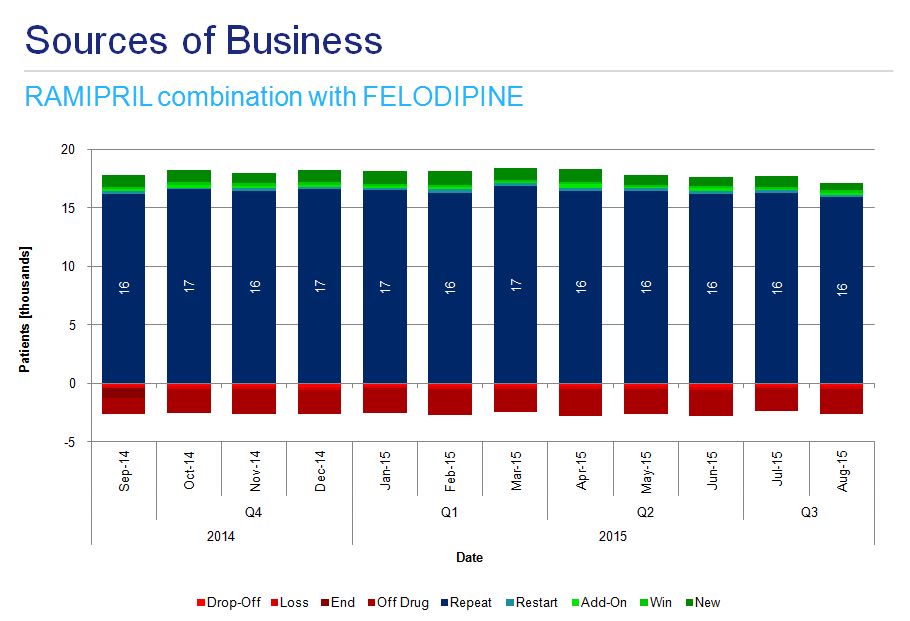
Analysis of instances of combination therapy among hypertensive patients indicates the most dynamic growth in the use of ramipril-amlodipine preparations (fig. 7a) and more stable usage of ramipril-felodipine and ramipril-hydrochlorothiazide (fig. 7b and 7c).
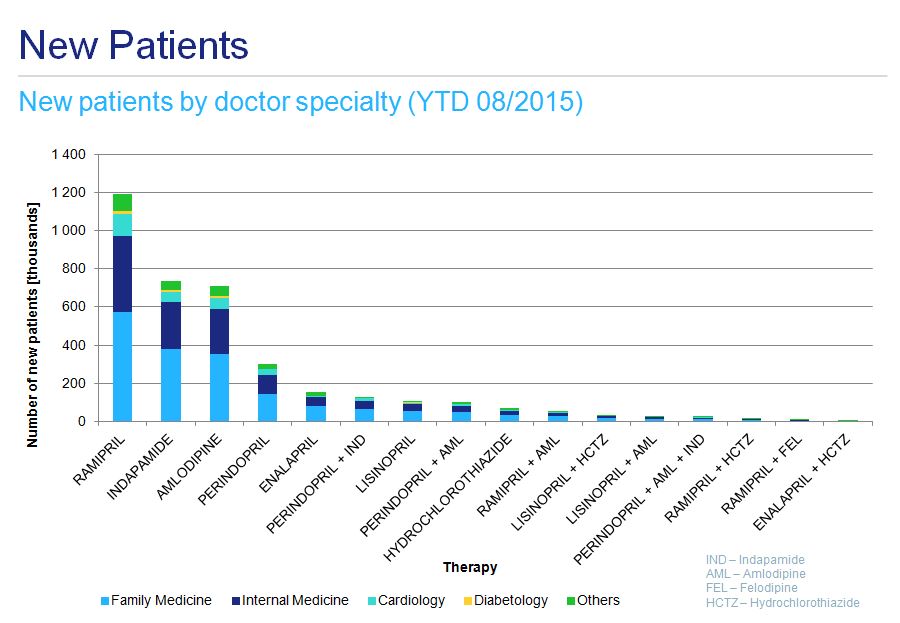
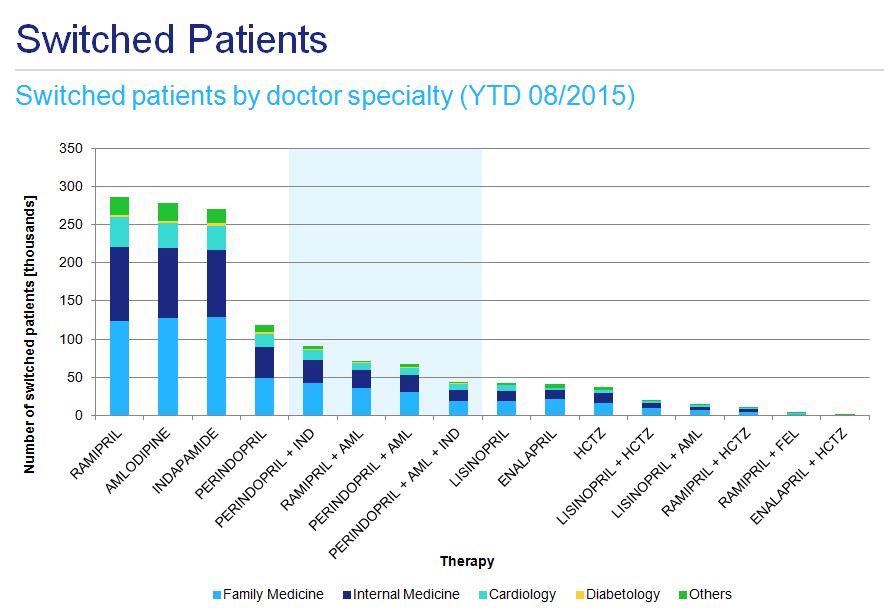
Both the initiation of hypertensive therapy and any subsequent changes are mainly prescribed by family medicine doctors and internal medicine specialists, with a less pronounced contribution from cardiologists (fig. 8 and fig. 9). This applies to ramipril monotherapy as well as fixed combination treatment.
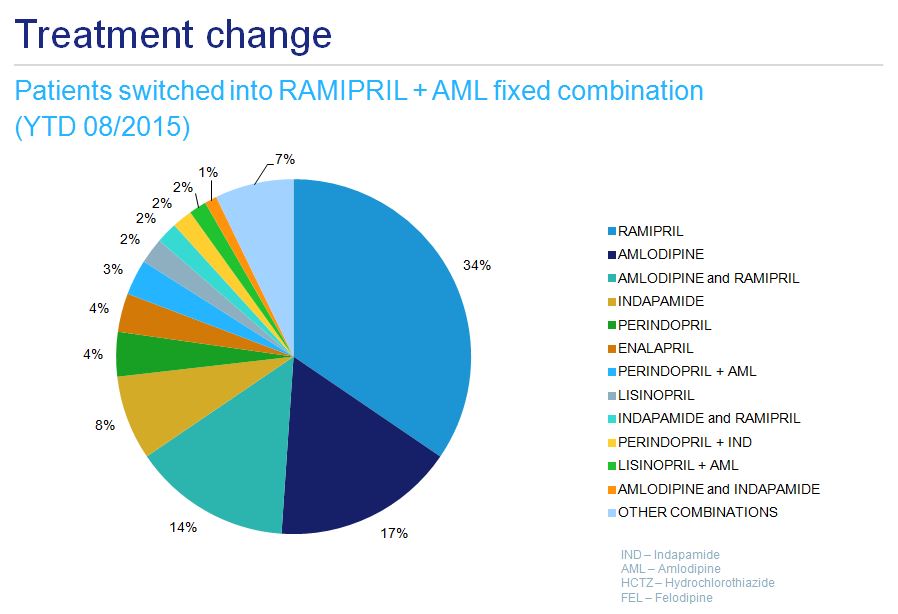
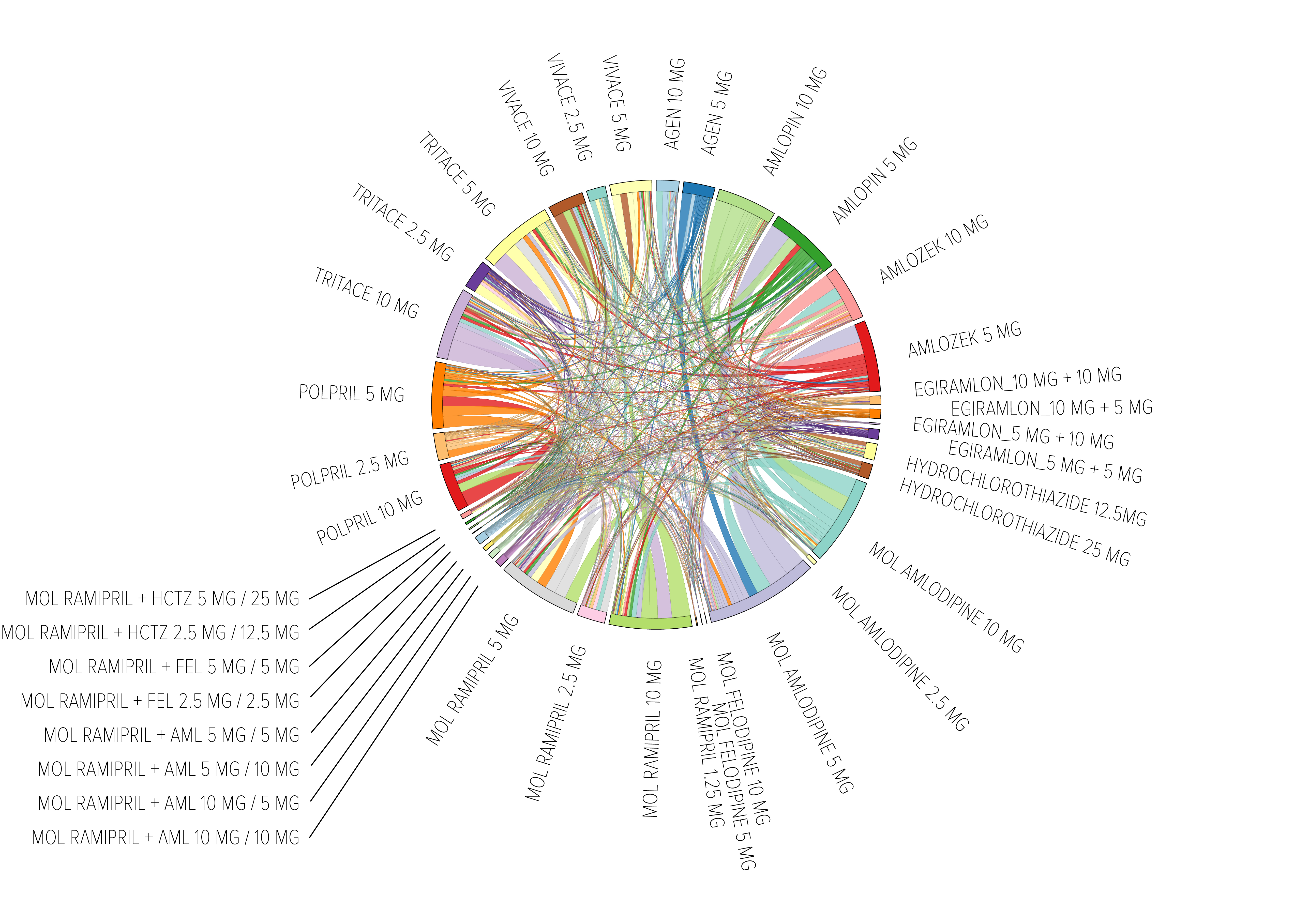
Owing to the multiple potential treatment options, treatment patterns in hypertension, even considered only at international names level, are very complex. They should be analyzed one by one with a certain degree of detail. For example: patients on a ramipril-amlodipine fixed combination would previously have been treated with ramipril alone, amlodipine alone, or a loose combination of these 2 drugs, or with another hypertension treatment (i.e. indapamide, perindopril, enalapril, perindopril-indapamide combination, lisinopril or others) (fig. 10). If one tries to consider all changes in ramipril-treated patients the picture becomes very complex (fig.11).
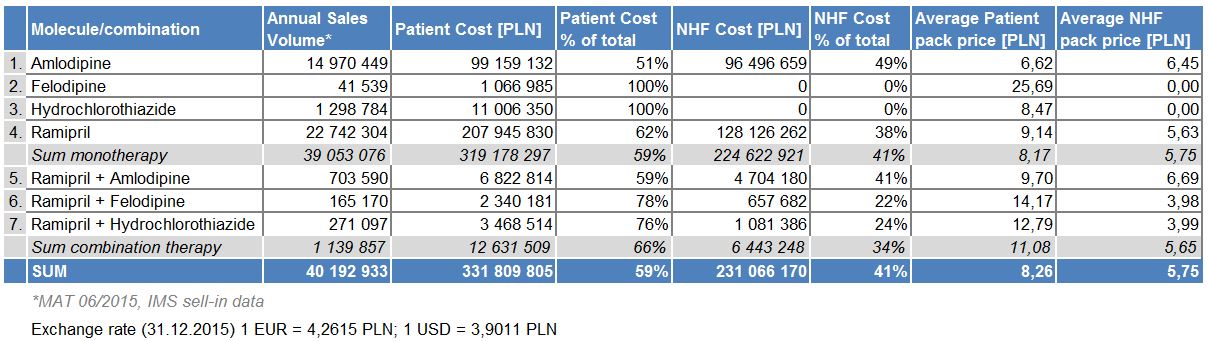
Cost analysis over a one-year time period indicates more than 25 times higher the contribution of costs from monotherapy than the costs of fixed-dose combinations. For monotherapy treatments, patient co-payments are almost 45% higher than the cost covered by the National Health Fund (NHF). For fixed combination therapies, patient contributions are even higher in percentage terms (tab. 1). The total treatment cost for this sub-group of anti-hypertensive drugs is substantial, mainly due to the high prevalence of the disease (the drugs themselves are relatively inexpensive). In case of the two most popular molecules, ramipril and amlodipine combination therapy is more expensive on average for both the patient and NHF than in monotherapy, which might explain its limited popularity. However, when loose combination of these products is considered, savings are available on both sides. The same is true for molecules not reimbursed in monotherapy, felodipine and hydrochlorothiazide, but their popularity is limited compared to the two leaders.
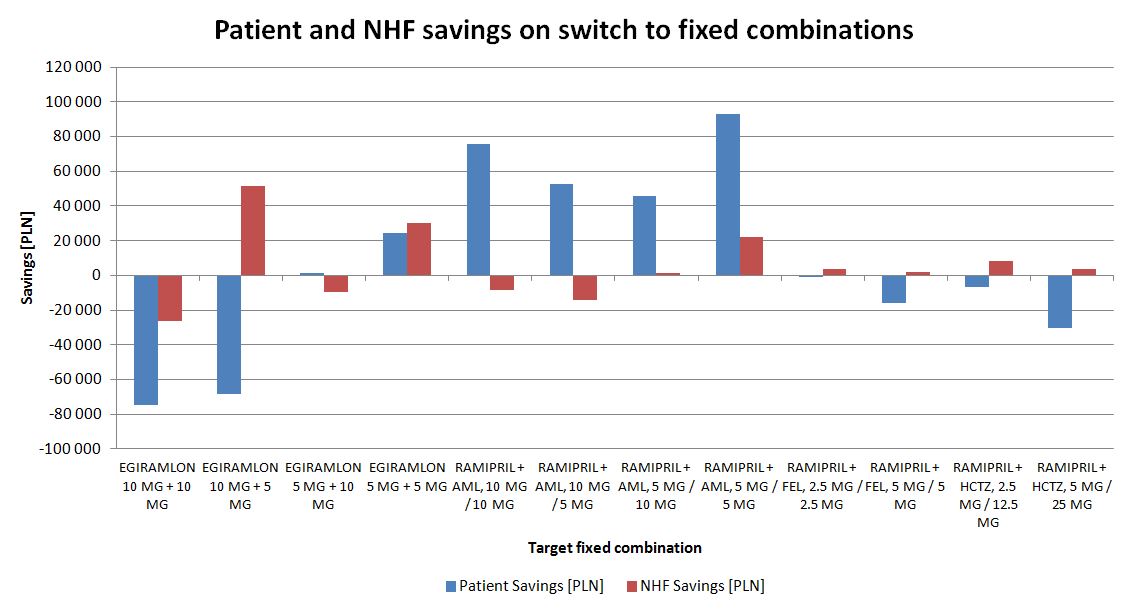
Savings are available for both patients and the public payer arising from treatment switches to fixed dose combination therapy in ramipril and ramipril- related therapy (fig. 12). A cost increase is observed mainly when the dose of medication is also increased (in addition to unit costs).
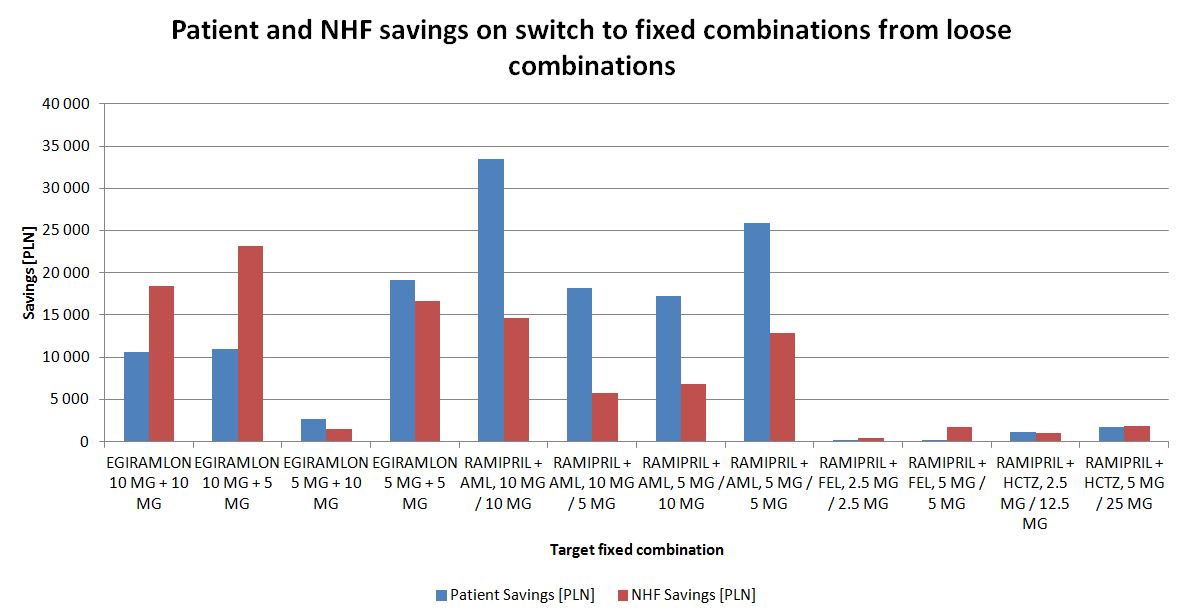
If only switches from loose combinations to fixed-dose combinations are considered, only savings are observed (fig. 13.). The scale of the savings however is small compared to the total spending on hypertensive drugs.
Figure 13. Savings from NHF and patients perspectives after switches from loose antihypertensive combinations to fixed dose combinations containing the same substances
Discussion
Due to the scale of the phenomenon, effective and cos-effective therapy of hypertensive patients is needed by the society and payers.
Improved allocation of scarce healthcare resources in this therapy area, where so many reasonably priced medicines are available, would allow for funds to be reserved for the more complicated cases, while meeting the aims of primary prevention programmes. All these considerations should be made in a country- specific context reflecting specific healthcare conditions.
Similar studies on fixed-dose combination anti-hypertensive products, with reviews of the associated economics, were performed in neighbouring countries - Germany [11], and Ukraine [12] - leading to similar conclusions.
Longitudinal, patient centric databases serve as an ideal source of real-world information. In our study, we have analyzed treatment patterns in hypertension on a large, representative sample of the Polish population. We put special emphasis on fixed-dose combination products containing ramipril, and ramipril itself, as the most commonly used drug from the ACEI group. Medications from the same group have also been assessed previously in Poland, either from clinical perspective [13,14] or from economic [15], healthcare sector management [16] viewpoints.
There are limitations of our study, as follows:
- Lack of effectiveness measures, even as secondary end points; it can be mitigated by including the results from large cohort studies performed in Poland
- Compliance and persistence not measured; a longer time horizon would be more appropriate to meet this objective
- Analysis includes only the cost of drugs, while other treatment cost components are missing. It can be argued however that these are irrelevant to this study and outside of its scope
- Only selected hypertension treatment options included. Broadening the scope would require a modified approach but it is possible to be performed with the same methodology (for selected medications, group of drugs or for all anti-hypertensive products)
Further research and analysis are needed to respond to these limitations . On the other hand, however, to our knowledge this is the largest study to date of hypertension therapies using RWE datasets in Poland.
Conclusions
Anti-hypertensive therapy patterns are very complex among Polish patients. They can be analyzed in detail with RWE databases like IMS LRx. It is the only option to interpret patients behavior as well as doctors prescribing habits longitudinally in their real practice settings.
Analysis of the market leader in the ACEI group, ramipril and ramipril-based drugs indicate that a wider use of fixed-dose combinations versus loose combinations offers cost savings for both patients and the public payer. Savings also arise when patients are switched from ramipril monotherapy to fixed-dose combinations, especially when the dose is not increased.
Therefore a switch to fixed-dose combinations would be justified not only from clinical (where indication in individual patient exists) but also from the economic perspective. It is in line with current guidelines on anti-hypertensive treatment.
Ethical statement
No conflict of interest declared. No funding received. All authors are employed by IMS Health.
- Rawlins M, “The Testimono”, 2008
- Garrison LP Jr, Neumann PJ, Erickson P, Marshall D, Mullins CD, Using real-world data for coverage and payment decisions: the ISPOR Real-World Data Task Force report, Value Health, 2007 Sep-Oct; 10(5): 326-38
- Wierzbicka N, Jahnc-Rozyk K, The evolving landscape for Real World Evidence in Poland: physicians’ perspective, JHPOR.2015.1.3: 15-33; DOI: 10.7365
- Available from: www.aotm.gov.pl cited 24.12.2015
- Szkultecka-Dębek M, Drozd M, Real world data guidelines - current status review, JHPOR, 2015, 1, 10-14 / JHPOR.2015.1.2; DOI: 10.7365
- Willke RJ, Mullins CD, “Ten commandments” for conducting comparative effectiveness research using “real-world data”. Supplement to Journal of Managed Care Pharmacy JMCP, November/December 2011, Vol. 17, No. 9-a
- Roche N, Reddel H, Martin R, Brusselle G, Papi A, Thomas M, Postma D, Thomas V, Rand C, Chisholm A, Price D, Respiratory Effectiveness Group: Quality standards for real-world research. Focus on observational database studies of comparative effectiveness, Ann. Am. Thorac. Soc. 2014 Feb; 11 Suppl 2: S99-104; doi: 10.1513/ AnnalsATS.201309-300RM
- von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP, Strengthening the reporting of observational studies in epidemiology (STROBE) statement: guidelines for reporting observational studies, BMJ, 2007 Oct 20, 335(7624), 806–808; doi: 10.1136/ bmj.39335.541782.AD
- Bandosz P. et al., Decline in mortality from coronary heart disease in Poland after socioeconomic transformation: modelling study, BMJ, 2012;344:d8136 doi: http://dx.doi.org/10.1136/bmj.d8136
- Grodzicki T. et al., Zasady postępowania w nadciśnieniu tętniczym: Wytyczne Polskiego Towarzystwa Nadciśnienia Tętniczego oraz Kolegium Lekarzy Rodzinnych w Polsce Nadciśnienie Tętnicze, 2008, Vol. 12, No. 5: 317-342
- Keiser E. et al., A database analysis to evaluate the risk of cardiovascular events in hypertensive patients being treated with either the single pill combination of valsartan and amlodipine or the respective free combination, Journal of Hypertension, June, 2011, vol. 29
- Mishchenko O, Iakovlieva L, Adonkina V, Pharmacoeconomic evaluation of fixed-dose triple combination for antihypertensive therapy in Ukraine, JHPOR, 2014, 1, 66-75, DOI: 10.7365/JHPOR.2014.5.8
- Filipiak K et al., Ramipril and amlodipine - new fixed-dose combination in antihypertensive treatment, Nadciśnienie Tętnicze, 2012, Vol. 16, No. 2: 105-119
- Widecka K. et al., Clinical effectiveness of a fixed-dose combination therapy with ramipril plus felodipine ER compared to monotherapy with ramipril or felodipine ER or to a fixed-dose combination therapy with verapamil SR and trandolapril or amlodipine and valsartan in treatment of primary hypertension - systematic review, Nadciśnienie Tętnicze, 2011, Vol 15, No. 1: 13-20
- Hermanowski T, Jaworski R, Czech M, Pachocki R, Ocena kosztów i efektów leczenia hipotensyjnego przy użyciu dwóch różnych inhibitorów konwertazy angiotensyny: peryndoprylu i enalaprylu, Farmakoekonomika 4/2001; 2-12
- Hermanowski T, Borowiec Ł, Faluta T., How to optimize public spending on antihypertensive treatment in Poland - an example of rationalization analysis, JHPOR, 2014, 2, 78-87 DOI: 10.7365/JHPOR.2013.4.9