System of medicinal maintenance in Russia
-
Copyright
© 2012 PRO MEDICINA Foundation, Published by PRO MEDICINA Foundation
User License
The journal provides published content under the terms of the Creative Commons 4.0 Attribution-International Non-Commercial Use (CC BY-NC 4.0) license.
Authors
| Name | Affiliation | |
|---|---|---|
Roza Yagudina |
Head of Department of Organization of Medicinal Provision and Pharmacoeconomics of First Moscow state medical university named I.M. Sechenov, Moscow
|
Medicinal maintenance
Medicinal maintenance of the population is one of the primary problems in any system of public health services all over the world. The Russian Federation isn't an exception and by the current moment it possesses an advanced system of medicinal maintenance that includes several market spheres which are commercial and hospital markets and the market of preferential medicinal maintenance.
Registration
The first stage of the circulation of pharmaceuticals that directly concerns medicinal maintenance is registration of the medical product as it separates biologically active substances from pharmaceuticals themselves. Since 2010 responsibility for the State Marketing Authorization have been allocated to Department of State Marketing Authorization of Medical Products that is a department of Ministry of Healthcare of Russia. At present there is the State Register of Medical Products that contains all the pharmaceuticals registered in the country.
According to the data obtained on the 3rd of February 2012 there are 17 161 registered in Russia medical products process by inland and foreign manufacturers; about 90% of them are pharmaceutical, while 10% are pharmaceutical substances used for production of pharmaceuticals. Whereas in 2011 Register of Medical Products was contained over 18 900 items, and in 2010 – over 19 300, that showed gradual decrease of quantity of registered pharmaceuticals and annual accurate reconsideration of the list of the registered drugs.
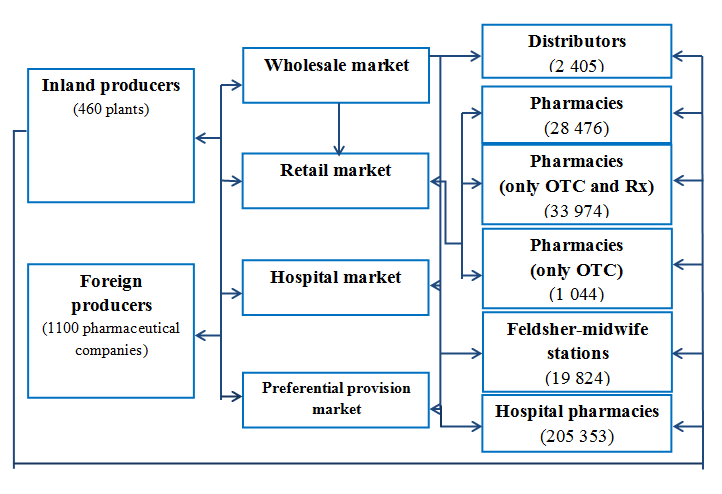
Russian pharmaceutical market is a complex and advanced structure that can satisfy the requirements of population and government in medical products by means of its organization and multitude of participants. The pharmaceutical market itself consists of the four main subdivisions that are wholesale market, retail market, hospital market and preferential provision market. Manufacturing and distributing are the two main states of circulation of medical products in the system of medicinal maintenance and they are carried out by variety of market insiders.
Manufacturing
According to the data of 2011 in Russia there are over 1 500 functional pharmaceutical manufacturers presented by 460 inland producers and about 1 100 foreign producers.
Distribution
The primary market participants that take place in the process of drugs dispensing are pharmaceutical distributers, pharmacies and medical and preventive treatment facilities. Total quantity of pharmaceutical distributors was about 2 400 in 2011. Pharmacies in Russia are presented with several types that are pharmacies that has license for selling all kinds of medicines (OTC, Rx and narcotic drugs), pharmacies that are prohibited to sell narcotic drugs but can sell OTC and Rx medicines and pharmacies that are allowed to sell only OTC medicines. Total quantity of licensed pharmacies in Russia in 2011 was about 64 855; about 44% them were able sell all types of drugs, 52% - only OTC and Rx drugs and less than 1% - only OTC drugs. In countryside and under-populated areas drugs dispensing effects by means of over 200 000 preventive treatment facilities as well those also include about 19 824 feldsher-midwife stations that have license for drug dispending. The structure and the main participants of Russian pharmaceutical market are presented in the figure 1.
Market volumes
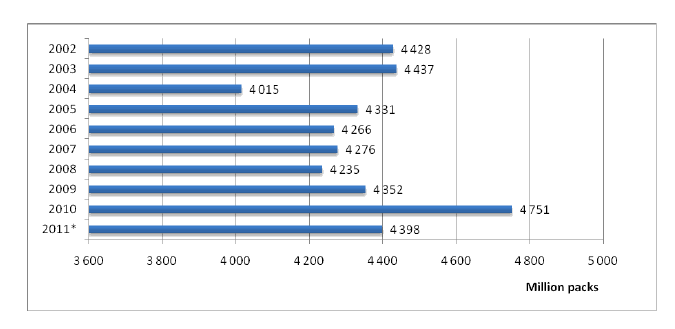
According to the data of Market Research Center “Pharmexpert” total market volume in natural units (in packs of pharmaceuticals) shows annual increase in the last years; it is presented in the figure 2.
* Data does not include preferential medicinal maintenance market segment.
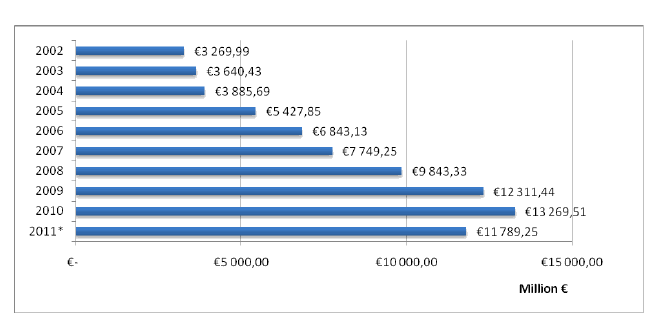
Dynamics of drug consumption in natural units shows stable annual market size. Distribution among the primary market spheres that are commercial (retail) and hospital markets and the market of preferential medicinal maintenance shows annual domination of commercial market segment. The size of retail market segment in 2010 have been about 4 395 000 000 packs (92% of the total market size), the size of the hospital market - 265 000 000 packs (6%) and preferential medicinal maintenance market - 91 800 000 packs (2%). Market volumes in financial units (Euro) also present annual market growth; it is presented on figure 3.
* Data does not include preferential medicinal maintenance market segment.
Distribution of market volumes among the primary market spheres in financial units has been showing high annual priority and growth of commercial (retail) market since 2005. The size of retail market segment in 2010 has been about € 10 258 314 440 (77% of the total market size), the size of the hospital market - € 942 807 500 (7%) and preferential medicinal maintenance market - € 2 068 391 900 (16%).
As can be seen from the above there is shown stable growth of Russian pharmaceutical market in natural and financial units since 2005 till nowadays.
Medical products consumption
Using the data of Market Research Center "Pharmexpert" there were composed tops of medical products that were the most frequently used in 2011 in Russia in natural and economical units. Medical products presented with international nonproprietary names (INN) were ranged according to the consumption volumes in different market segments. The most frequently used drug in natural units in retail market is Xylometazoline; in hospital market - sodium chloride; in preferential medicinal maintenance market – human genetically engineered insulin. Top-5 pharmaceuticals in natural units according to the market segment value are presented in table 1.
Table 1. Top-5 pharmaceuticals in natural units in Russia (2011)
| Commercial (retail) market | Hospital market | Preferential medicinal maintenance market |
| Xylometazoline | Sodium chloride | Human genetically engineered insulin |
| Pancreatine | Ceftriaxone | Blood-coagulation factor VIII |
| Polyvitamin+Multimineral | Heparin sodium | Insulin glargine |
| Nimesulide | Levofloxacin | Epoetin alfa |
| Methylphenylthiomethyl-dimethylaminomethyl-hydroxybromindol-ethyl carboxylat | Meropenem | Interferon beta-1b |
Medical products in financial units were either arranged according to the volume of the consumption. The most frequently used drug in financial units in retail market is combination of Polyvitamin and Multimineral: in hospital market - sodium chloride; in preferential medicinal maintenance market – human genetically engineered insulin. Top-5 pharmaceuticals in natural units according to the market segment value are presented in table 2.
Table 2. Top-5 pharmaceuticals in financial units in Russia (2011)
| Commercial (retail) market | Hospital market | Preferential medicinal maintenance market |
| Polyvitamin+Multimineral | Sodium chloride | Imatinib |
| Methylphenylthiomethyl-dimethylaminomethyl-hydroxybromindol-ethyl carboxylat | Meropenem | Rituximab |
| Xylometazoline | Oxaliplatin | Blood-coagulation factor VIII |
| Phospholipids | Docetaxel | Bortezomib |
| Pancreatine | Ceftriaxone | Glatiramer acetate |
System of preferential medicinal maintenance
Nowadays in Russia there exists a system of Government Reimbursement for the preferential patients that includes a number of federal and regional programs.
In the beginning of 2005 there have been started a program of Additional Pharmacological Support (APS) that had the reimbursement drug list in the base and gave an opportunity to improve quality and accessibility of preferential provision of medicines for vulnerable social group. There was developed List of Prescription Medicines in Additional Drug Supplement in the National Social Care during the time of the program realization; it was main characteristics were organization according to the INN, validation of maximum sale price. These characteristics help to decrease disproportion in preferential medicinal maintenance market. The main issues of APS are personalization of the given pharmacological support, normalization of financing of preferential patients and organization of data connection and data reporting in reimbursement pharmacological and medicinal support.
In 2008 program called "7 diseases" was separated from the APS program. It is a program of ambulatory medicine reimbursement of patients with the rare and high-cost diseases that are hemophilia, cystic fibrosis, pituitary dwarfism, Gaucher disease, multiple sclerosis, myeloleukemia and organs and (or) tissue transplantation. Therefore this new federal program become one of the priory reimbursement programs of drug supply that is responsible for government financing of preferential patients by the means of centralized purchases of medical products. Total market volume in preferential medicinal maintenance segment of "7 diseases" in 2011 on natural units was about 2 195 300 packs, and in financial units – about € 804 132 900. Distribution among the diseases is presented in table 3.
Table 3. Market volumes of "7 diseases" sphere
| Disease | Natural units | Financial units |
| Myeloleukemia | 2 195 334 | € 804 132 926,81 |
| Hemophilia | 262 462 | € 345 840 925,24 |
| Multiple sclerosis | 1 044 537 | € 185 187 052,03 |
| Transplantations | 375 166 | € 178 995 858,21 |
| Gaucher disease | 311 801 | € 37 797 795,76 |
| Cystic fibrosis | 14 952 | € 27 222 176,01 |
| Pituitary dwarfism | 115 656 | € 23 410 373,91 |
Perspectives of prescription drug insurance
The first idea of prescription drug insurance appeared in 2008 but there was no opportunity to develop it at that moment. According to the Minister of health and social development of the Russian Federation Tatyana Golikova now there is a perspective to develop the system of prescription drug insurance by 2015. This system is about vital and necessary medical products that are presented in the list of the Vital and Essential Medicines. Two approaches are considered: insurance - more difficult both more expensive; and compensation. At the first stage the main aim is to provide with medicines those for whom it's necessary and first of all to solve the regional problem. But in general it is a question of providing with medical care of all the patients in the out-patient sphere.
There is either a system called Population Drug Coverage that works by the means of regional purchases of medical products. The sources of financing nowadays are different level budgets (regional and municipal), budget of the Compulsory Health Insurance fund and personal means of population.
Conclusion
As can be seen from the above Russian pharmaceutical market is a continuously developing and growing structure that includes a multitude of participants in different levels of organization. Annual market researches illustrate increase of volumes of all of the market spheres either in economical or in natural values. Therewith the perspective of prescription drug insurance might become the new forcer of market development and is likely to redefine the whole system of medicinal maintenance in Russia.